Quantitative MRI of organspecific vascular permeability and immune cell dynamics by combination with mass spectrometric imaging
Principal investigators: Cornelius Faber, Uwe Karst
Project number: CRC 1450 B02
Project term: 01/2021–12/2028
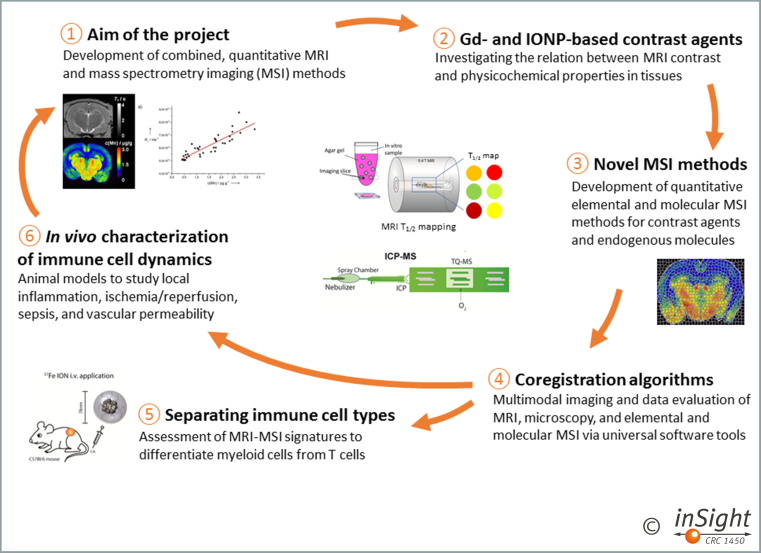
The aim of this project is to develop combined in vivo MRI and ex vivo mass spectrometry imaging (MSI) methods including both, laser ablation-inductively coupled plasma-mass spectrometry imaging (LA-ICP-MSI) and matrix-assisted laser desorption/ionisation mass spectrometry imaging (MALDI-MSI) (1). MSI shall be used to develop fully quantitative MRI methods (2) as part of our comprehensive multiscale imaging strategy (2, 3, 4) to analyse immune responses in disease models and patients in vivo (5).
The overarching question is how quantitative MR parameters depend on elemental and molecular patterns in tissues, and how this dependence evolves during inflammatory responses (6).
Team
Principal investigators
- Faber, Cornelius, Univ.-Prof. Dr. rer. nat.
Department of Radiology - Karst, Uwe, Univ.-Prof. Dr. rer. nat.
Institute of Inorganic and Analytical Chemistry
Project members
- Hiddeßen, Lena
Institute of Inorganic and Analytical Chemistry -
Sivaji, Surya
Department of Radiology
Publications
The names of the principal investigators in our network have been bolded. Publications released prior to 2021, when funding for our network commenced, represent previous project-related work.
2024
| Hoffmann E, Masthoff M, Kunz WG, Seidensticker M, Bobe S, Gerwing M, Berdel WE, Schliemann C, Faber C, Wildgruber M. Multiparametric MRI for characterization of the tumour microenvironment. Nat Rev Clin Oncol 2024;21: 428-448. Abstract |
| Kronenberg K, Hoffmann E, Hiddeßen L, Maus B, Gerwing M, Faber C, Sperling M, Wildgruber M, Karst U. P-based referencing for correcting tissue artifacts in laser ablation-inductively coupled plasma-mass spectrometry imaging of cancer samples. Metallomics 2024;16Abstract |
| Rivas Aiello MB, Kirse TM, Lavorato GC, Maus B, Maisuls I, Kuberasivakumaran S, Ostendorp S, Hepp A, Holtkamp M, Winkler EL, Karst U, Wilde G, Faber C, Vericat C, Strassert CA. Superparamagnetic Nanoparticles with Phosphorescent Complexes as Hybrid Contrast Agents: Integration of MRI and PLIM. Small Science 2024Abstract |
| Schache D, Peddi A, Nahardani A, Faber C, Hoerr V. Corrections for Rabi oscillations in cardiac chemical exchange saturation transfer MRI under the influence of very short preparation pulses. NMR Biomed 2024;37: e5081. Abstract |
| Wilken E, Havlas A, Masthoff M, Moussavi A, Boretius S, Faber C. Radial compressed sensing imaging improves the velocity detection limit of single cell tracking time-lapse MRI. Magn Reson Med 2024;91: 1449-1463. Abstract |
2023
| Armstrong M, Wilken E, Freppon F, Masthoff M, Faber C, Xiao D. Dynamic cell tracking using time-lapse MRI with variable temporal resolution Cartesian sampling. Magn Reson Med 2023;90: 2443-2453. Abstract |
| Elinkmann M, Reuter Sa, Holtkamp M, Heuckeroth S, Köhrer A, Kronenberg K, Sperling M, Rubner O, Quarles CD, Hippler M, Karst U. Improving detection thresholds and robust event filtering in single-particle and single-cell ICP-MS analysis. J. Anal. At. Spectrom. 2023Abstract |
| Heuckeroth S, Behrens A, Wolf C, Fütterer A, Nordhorn ID, Kronenberg K, Brungs C, Korf A, Richter H, Jeibmann A, Karst U, Schmid R. On-tissue dataset-dependent MALDI-TIMS-MS(2) bioimaging. Nat Commun 2023;14: 7495. Abstract |
| Hoffmann E, Gerwing M, Krähling T, Hansen U, Kronenberg K, Masthoff M, Geyer C, Höltke C, Wachsmuth L, Schinner R, Hoerr V, Heindel W, Karst U, Eisenblätter M, Maus B, Helfen A, Faber C, Wildgruber M. Vascular response patterns to targeted therapies in murine breast cancer models with divergent degrees of malignancy. Breast Cancer Res 2023;25: 56. Abstract |
| Hoffmann E, Schache D, Höltke C, Soltwisch J, Niland S, Krähling T, Bergander K, Grewer M, Geyer C, Groeneweg L, Eble JA, Vogl T, Roth J, Heindel W, Maus B, Helfen A, Faber C, Wildgruber M, Gerwing M, Hoerr V. Multiparametric chemical exchange saturation transfer MRI detects metabolic changes in breast cancer following immunotherapy. J Transl Med 2023;21: 577. Abstract |
| Kronenberg K, Werner J, Bohrer P, Steiger K, Buchholz R, von Bremen-Kühne M, Elinkmann M, Paprottka PM, Braren RF, Lohöfer FK, Karst U. Simultaneous quantification of Gadoxetic acid and Cisplatin in hepatocellular carcinomas using laser ablation-inductively coupled plasma-mass spectrometry. Metallomics 2023;15Abstract |
2022
| Gerwing M, Hoffmann E, Kronenberg K, Hansen U, Masthoff M, Helfen A, Geyer C, Wachsmuth L, Höltke C, Maus B, Hoerr V, Krähling T, Hiddeßen L, Heindel W, Karst U, Kimm MA, Schinner R, Eisenblätter M, Faber C, Wildgruber M. Multiparametric MRI enables for differentiation of different degrees of malignancy in two murine models of breast cancer. Front. Oncol. 2022;12Abstract |
| Schwarz C, Buchholz R, Jawad M, Hoesker V, Terwesten-Solé C, Karst U, Linsen L, Vogl T, Hoerr V, Wildgruber M, Faber C. Fingerprints of Element Concentrations in Infective Endocarditis Obtained by Mass Spectrometric Imaging and t-Distributed Stochastic Neighbor Embedding. ACS Infect Dis 2022Abstract |
2021
| Gerwing M, Krähling T, Schliemann C, Harrach S, Schwöppe C, Berdel AF, Klein S, Hartmann W, Wardelmann E, Heindel WL, Lenz G, Berdel WE, Wildgruber M. Multiparametric Magnetic Resonance Imaging for Immediate Target Hit Assessment of CD13-Targeted Tissue Factor tTF-NGR in Advanced Malignant Disease. Cancers (Basel) 2021;13Abstract |
| Karlas A, Masthoff M, Kallmayer M, Helfen A, Bariotakis M, Fasoula NA, Schäfers M, Seidensticker M, Eckstein H-H, Ntziachristos V, Wildgruber M. Multispectral optoacoustic tomography of peripheral arterial disease based on muscle hemoglobin gradients-a pilot clinical study. Ann Transl Med 2021;9: 36. Abstract |
| Masthoff M, Freppon FN, Zondler L, Wilken E, Wachsmuth L, Niemann S, Schwarz C, Fredrich I, Havlas A, Block H, Gerwing M, Helfen A, Heindel W, Zarbock A, Wildgruber M, Faber C. Resolving immune cells with patrolling behaviour by magnetic resonance time-lapse single cell tracking. EBioMedicine 2021;73: 103670. Abstract |
| Spieth L, Berghoff SA, Stumpf SK, Winchenbach J, Michaelis T, Watanabe T, Gerndt N, Düking T, Hofer S, Ruhwedel T, Shaib AH, Willig K, Kronenberg K, Karst U, Frahm J, Rhee JS, Minguet S, Möbius W, Kruse N, von der Brelie C, Michels P, Stadelmann C, Hülper P, Saher G. Anesthesia triggers drug delivery to experimental glioma in mice by hijacking caveolar transport. Neurooncol Adv 2021;3: vdab140. Abstract |
2019
| Masthoff M, Buchholz R, Beuker A, Wachsmuth L, Kraupner A, Albers F, Freppon F, Helfen A, Gerwing M, Höltke C, Hansen U, Rehkämper J, Vielhaber T, Heindel W, Eisenblätter M, Karst U, Wildgruber M, Faber C. Introducing Specificity to Iron Oxide Nanoparticle Imaging by Combining (57)Fe-Based MRI and Mass Spectrometry. Nano Lett 2019;19: 7908-7917. Abstract |
2018
| Fingerhut S, Sperling M, Holling M, Niederstadt T, Allkemper T, Radbruch A, Heindel W, Paulus W, Jeibmann A, Karst U. Gadolinium-based contrast agents induce gadolinium deposits in cerebral vessel walls, while the neuropil is not affected: an autopsy study. Acta Neuropathol 2018;136: 127-138. Abstract |
| Kröger S, Niehoff A-C, Jeibmann A, Sperling M, Paulus W, Stummer W, Karst U. Complementary Molecular and Elemental Mass-Spectrometric Imaging of Human Brain Tumors Resected by Fluorescence-Guided Surgery. Anal Chem 2018;90: 12253-12260. Abstract |
| Lohöfer F, Hoffmann L, Buchholz R, Huber K, Glinzer A, Kosanke K, Feuchtinger A, Aichler M, Feuerecker B, Kaissis G, Rummeny EJ, Höltke C, Faber C, Schilling F, Botnar RM, Walch AK, Karst U, Wildgruber M. Molecular imaging of myocardial infarction with Gadofluorine P - A combined magnetic resonance and mass spectrometry imaging approach. Heliyon 2018;4: e00606. Abstract |
| Masthoff M, Gran S, Zhang X, Wachsmuth L, Bietenbeck M, Helfen A, Heindel W, Sorokin L, Roth J, Eisenblätter M, Wildgruber M, Faber C. Temporal window for detection of inflammatory disease using dynamic cell tracking with time-lapse MRI. Sci Rep 2018;8: 9563. Abstract |
2017
| Niehoff A-C, Klasen T, Schmidt R, Palmes D, Faber C, Karst U, Hadrian R. Investigating the Lymphatic System by Dual-Color Elemental Mass Spectrometry Imaging. Contrast Media Mol Imaging 2017;2017: 4035721. Abstract |

