Developmental biology: My research about the coronary vasculature

How does the coronary vasculature develop? What role does molecular signalling from the coronary vessels play in cardiovascular disease? These are the questions that underlie my research in the laboratory of Prof. Ralf Adams at the Max Planck Institute for Molecular Biomedicine in Münster. One of the reasons I am interested in these questions is because every year, almost four million people in Europe die because of cardiovascular disease; this makes up 45% of all deaths.
The coronary vasculature includes all the blood vessels that supply the cardiac muscle, the heart, with oxygen and nutrients. These vessels split off from the aorta and cover the heart like branches of a tree. Being able to understand the molecular mechanisms that govern this vasculature would allow scientists to develop new diagnostic methods and, hopefully, new therapies. When or even if this will be possible is unclear, but it is why I go in the lab every morning.
My scientific question
During my PhD in my hometown of Madrid, I was already very interested in the development and disease of the cardiovascular system. At the Centro Nacional de Investigaciones Cardiovasculares, in the laboratory of José Luis de la Pompa, I was interested in signalling pathways involved in cellular communication in the context of a particular type of heart disease. I studied the ventricles, the main chambers of the heart, which are responsible for pumping blood to the whole body. During this work, it called my attention that the ventricles of a certain genetically modified mouse line did not present blood vessels. It was this observation that brought me to the Max Planck Institute in Münster to study the biology of the coronary vasculature.
Currently, I study a particular signalling pathway in the coronary vasculature of adult mice called Eph-ephrin signalling. This signalling pathway is already known to play an important role in vascular formation early on, during the determination of veins and arteries, when the primitive blood vessels in the embryo have to decide whether to become an artery or a vein. But, what role these molecules play in the adult vasculature, especially in the coronary vasculature, are not yet deeply understood. This is the question I am trying to answer.
A look into my laboratory daily life
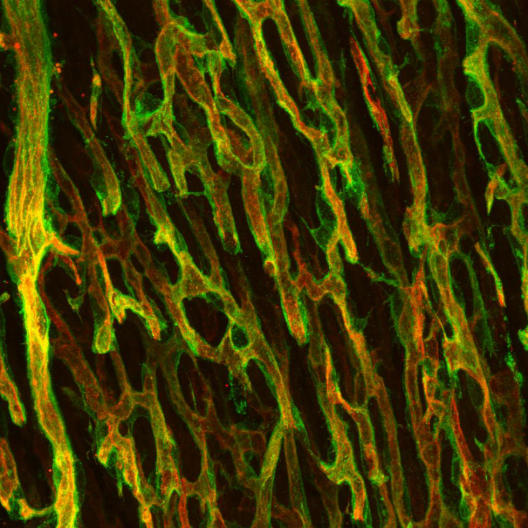
As a developmental biologist, I spend many hours in front of the microscope analysing the structure of the coronary vessels. I also analyse where and how strongly different molecular markers are expressed in the endothelial cells, the cells that form the inner lining of the blood vessels. Because blood vessels run deep in our organs, we have to cut into the tissue to reveal the inner structures. While histology is traditionally done using very thin sections of tissue that have been stained to study the different structures of different organs, this approach is not ideal for looking at blood vessels because thin sections do not allow for analysing the vascular bed’s complicated three-dimensional structure. If you think of a blood vessel as a pipe, when you cut through a very thin section of pipe all you can see is a circle.

Instead of using thin sections, in our laboratory we use some of the latest techniques, allowing us to analyse thicker sections of tissue that still have their spatial information intact. In that way, we do not see just a circle but the whole pipe, and even the “neighbours” around it. To obtain these sections, I work with a device called cryostat, which cuts sections out of a frozen block that contains the sample tissue. These sections are ten times thicker than the traditional histology thin sections. Although we call these cryostat-obtained sections “thick”, in reality the tissue sections still only measure 0.05 millimetres across. Once cut, we stick the tissue sections onto histology slides, the thin glass plates that can be put under the microscope for further analysis.
The whole process of preparing samples so they are ready to be sectioned under the cryostat takes one week. First, I fix the dissected tissue so that it maintains, as closely as possible, the properties it had in the living organism. To do this, the tissue is embedded in a sucrose-rich solution that will not form crystals when frozen, as this would destroy the structure of the sample. We call this process cryoprotection. Finally, to form the block containing the tissue that will eventually be cut into sections, I exchange the sucrose solution for a different one containing sucrose and gelatin, then I freeze it at -80°C. The resulting block is 1.5 cm wide, 1.5 cm long and 0.5 cm deep. Once frozen, I can use the cryostat to cut the block into sections and then place the sections onto slides for further analysis. The most common analysis technique that I, as molecular biologist, perform on the sections is immunohistochemistry. For this technique, I employ antibodies to stain different proteins so that I can study their expression, meaning where they are expressed and how much of the protein is present. At the end of the process, I use the microscope to obtain colorful, vibrant pictures of the coronary vasculature. These pictures are more than just stunning; they help me come a bit closer to understand my scientific questions.
Background information "Science in a way that everyone can understand"
This article is the result of a communication training for junior scientists. The participants learned techniques for writing an interesting, easily readable text. Subsequently, they wrote an article about their research that also non-experts should understand and translated it into English. The communication team of the Cells-in-Motion Cluster of Excellence initiated the project and supported the participants in individual coaching sessions. The English support office of the University of Münster helped to optimise the translations. The aims: Reflecting terms of content and language when editing their own research topic should benefit the participants in their communication with the public but also within the scientific community. They also gain experience in working with communication departments and photographers.
Further articles resulting from this project:
- How nerve cells communicate in the fear network – Guest contribution by Dr. Lena Goedecke, biologist in the research group of Prof. Hans-Christian Pape at the Cells-in-Motion Cluster of Excellence
- How the cells’ environment affects their migration: Guest article by Sargon Groß-Thebing, PhD student in the group of Prof. Erez Raz at the Cells-in-Motion Cluster of Excellence
- How do neuronal processes degenerate? Guest contribution by Dr. Svende Herzmann, biologist in the research group of Dr. Sebastian Rumpf at the Cells-in-Motion Cluster of Excellence

