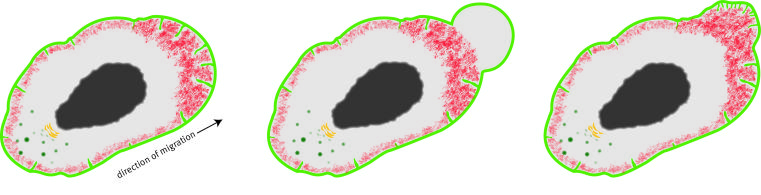
Cells use folds in their membrane for their motility
When an organism develops, large numbers of cells migrate from place to place in order to form tissue and organs in the right spots. In order to be able to move, some cells form protrusions in the form of blebs, which point out the direction in which they migrate. These protrusions look like balloons – but, in contrast to the rubber of a balloon, the outer layer of a cell, the cell membrane, cannot expand very much without breaking. How do the protrusions form then? Investigating the migration of primordial germ cells in zebrafish embryos, researchers at the Cells-in-Motion Cluster of Excellence at the University of Münster have discovered that certain folds in the membrane allow the cells to produce blebs. These folds are located inside the cell and they then unfold outwards during bleb formation. “We were able to show for the first time that these structures play a decisive role in the formation of the blebs,” says cell biologist Prof. Erez Raz, a group leader at the Cells-in-Motion Cluster of Excellence. This knowledge may also be useful for understanding the migration of other types of cells – for example, of certain cancer cells, which move in a similar way. The study has been published in the current issue of the journal “Developmental Cell”.
The detailed story:
Different types of cells migrate in different ways. Some cells develop protrusions in the form of blebs, in the direction of migration, which they use to move. These blebs are formed when pressure in the cytoplasm pushes the cell membrane outwards. In their study, the researchers examined this cell shape deformation in primordial germ cells that migrate within developing zebrafish embryos. Primordial germ cells migrate in the embryo from the place they are formed towards the region in the embryo where they then give rise to sperm and egg cells within the testis or the ovary. Primordial germ cells can change their shape and extend blebs within a few seconds. “We wanted to find out how the cells can change their shape so rapidly, and what is the source for the membrane ‘material’ required for their formation,” says cell biologist Mohammad Goudarzi, the lead author of the study.
The researchers took a close look at the cell membrane, and identified folds, which are located inside the cell. For the first time, they determined the role of these folds and their dynamic behaviour in a living organism. First, the researchers marked the cell membrane with a fluorescent protein in order to observe it under the microscope. Before the cell formed a bleb, folds could be clearly seen in the membrane. Once a bleb had formed, however, these structures could not be observed anymore – which led the researchers to assume that the folds unfolded and provide membrane material for bleb inflation.
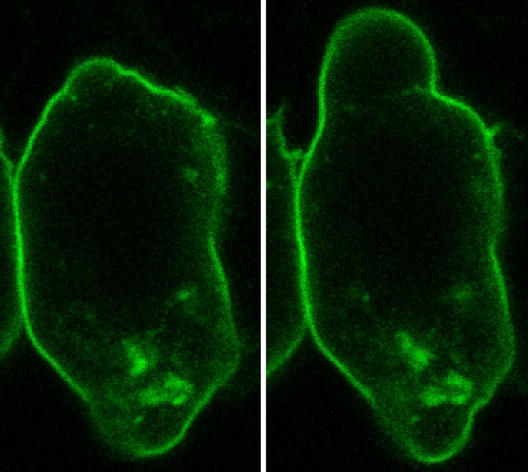
To study the dynamics of the unfolding process, the researchers additionally marked the membrane folds with certain proteins which tend to bind to curved membranes. The researchers were able to observe that those marker proteins were enriched on the front of the migrating cell, the position where the blebs formed. Moreover, inhibiting membrane unfolding resulted in inhibition of bleb formation and migration.
In addition, the researchers discovered that a component of the cell skeleton, the actin protein, was localized to the regions where the folds formed. Actin polymerization is regulated by the protein Cdc42 such that inhibiting its activity reduces the accumulation of actin. Using a confocal microscope, they were able to observe in the living organism that if there was less Cdc42 present, fewer folds were formed. As a result, fewer blebs were formed, the cells’ motility was reduced and, as a result, they no longer reached their target. “In this way, we were able to show that the formation of folds in the cell membrane is regulated by Cdc42 and that the folds are necessary for the formation of blebs and for the cell’s motility,” says Mohammad Goudarzi.
“Our latest findings relating to the question of how cells move could be of general interest – both in the context of physiological processes and also in diseases,” says Erez Raz, because not only primordial germ cells, but also other types of cell form blebs when they migrate: certain cancer cells, for example, when they invade healthy tissues. Protrusions are also formed on the cell’s surface during cell division or when cells die. In the future, the new findings could be tested in other cells and organisms, making them more general and potentially relevant for possible medical applications.
The study received funding from the Cells-in-Motion Cluster of Excellence at the University of Münster, from the Interdisciplinary Centre for Clinical Research (IZKF) at Münster University, as well as from the European Research Council and the German Research Foundation (DFG).
Original publication:
Goudarzi M, Tarbashevich K, Mildner K, Begemann I, Garcia J, Paksa A, M Reichman-Fried M, Mahabaleshwar H, Blaser H, Hartwig J, Zeuschner D, Galic M, Bagnat M, Betz T, Raz E. Bleb expansion in migrating cells depends on supply of membrane from cell surface invaginations. Dev Cell 2017, DOI 10.1016/j.devcel.2017.10.030. Abstract
Background:
A number of research groups from different disciplines at the Cells-in-Motion (CiM) Cluster of Excellence collaborated on the study. A team of biologists Ied by CiM Junior Research Group Leader Dr. Milos Galic developed special proteins for marking the folds of the cell membrane and following their dynamics. A team of biophysicists led by CiM Professor Timo Betz developed a special software for making accurate measurements of the fluorescent signals on the rapidly moving cell membrane.

