BODIPY-based self-assembled Structures
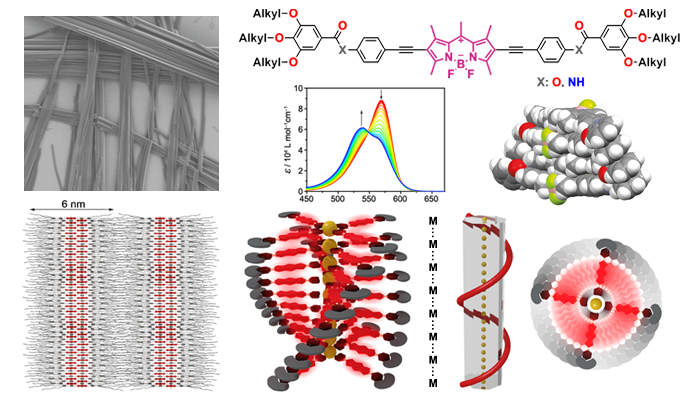
BODIPY dyes have attracted considerable attention in the past decades due to their excellent optical, photophysical and electronic properties, high chemical and physiological stability as well as ease in chemical functionalization. Their planar geometry anticipates facile stacking of the dipyrromethene core through aromatic interactions, which has been exploited to construct various supramolecular structures in solution and the solid state. Our group currently focuses on the design of hydrophobic and amphiphilic BODIPY dye molecules and understanding the fundamentals of their self-assembly processes in solution and the solid state. For instance, various BODIPY dye aggregates with alternated chromophore arrangement as well as H- and J-type excitonic coupling have been thoroughly investigated in our group. Controlling the molecular arrangement is key to understanding the functionalities of this class of fluorescent dyes, yielding promising functional materials for optoelectronic or biomedical applications.
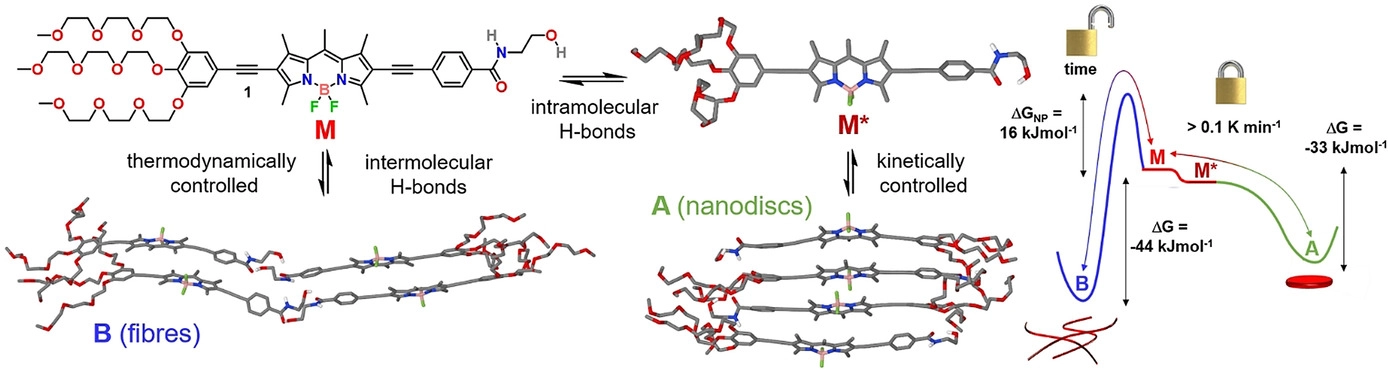
Pathway and Length Control of Supramolecular Polymers in Aqueous Media via a Hydrogen Bonding Lock
I.Helmers, G. Ghosh, R. Q. Albuquerque, G. Fernández
Angew. Chem. Int. Ed., 2021, 60, 4368–4376
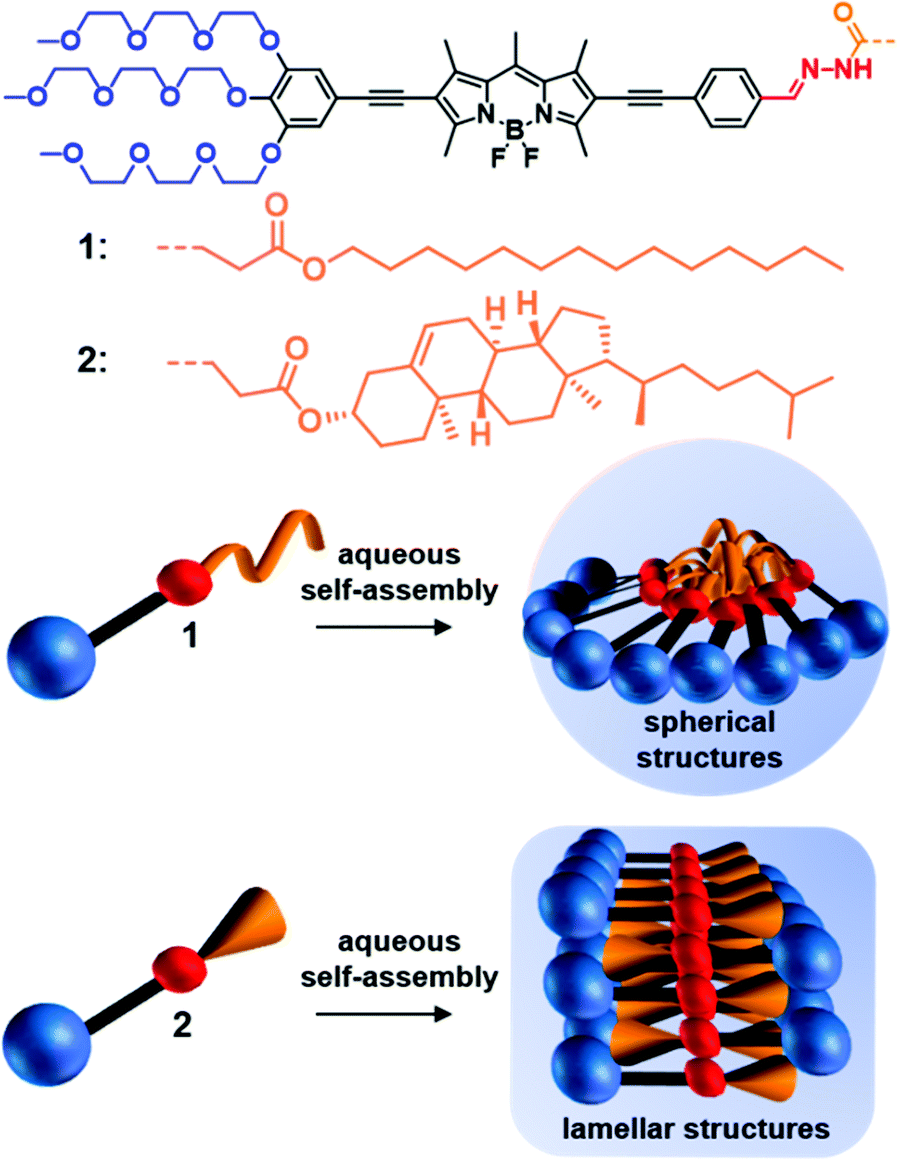
Hydrophobic domain flexibility enables morphology control of amphiphilic systems in aqueous media
I. Helmers, N. Bäumer, G. Fernández
Chem. Commun., 2020, 56, 13808-13811
DOI: 10.1039/D0CC06173E
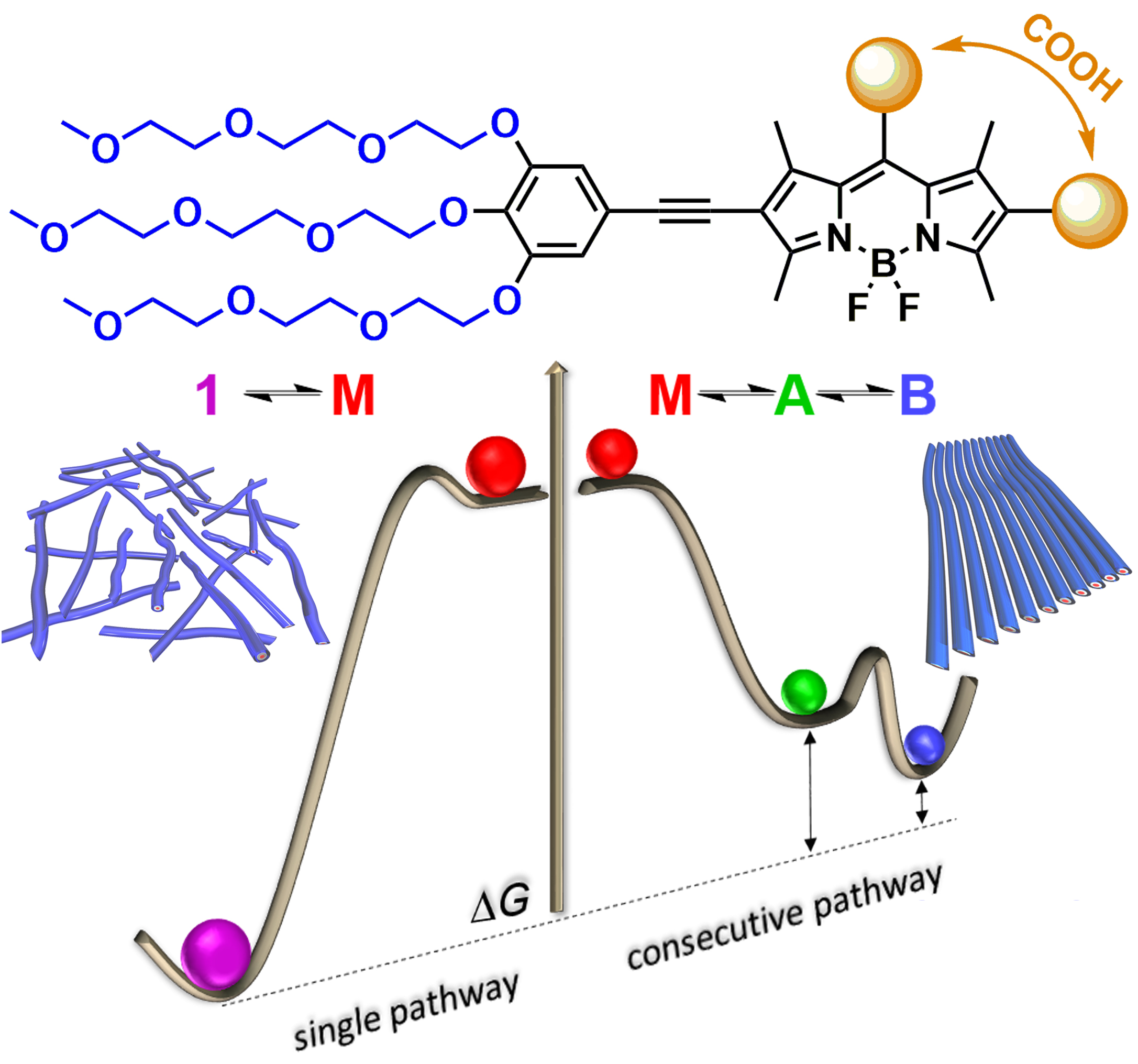
Synergistic repulsive interactions trigger pathway complexity
I. Helmers, M. Niehues, K. K. Kartha, B. J. Ravoo, G. Fernández
Chem. Commun., 2020, 56, 8944-8947
DOI: 10.1039/D0CC03603J
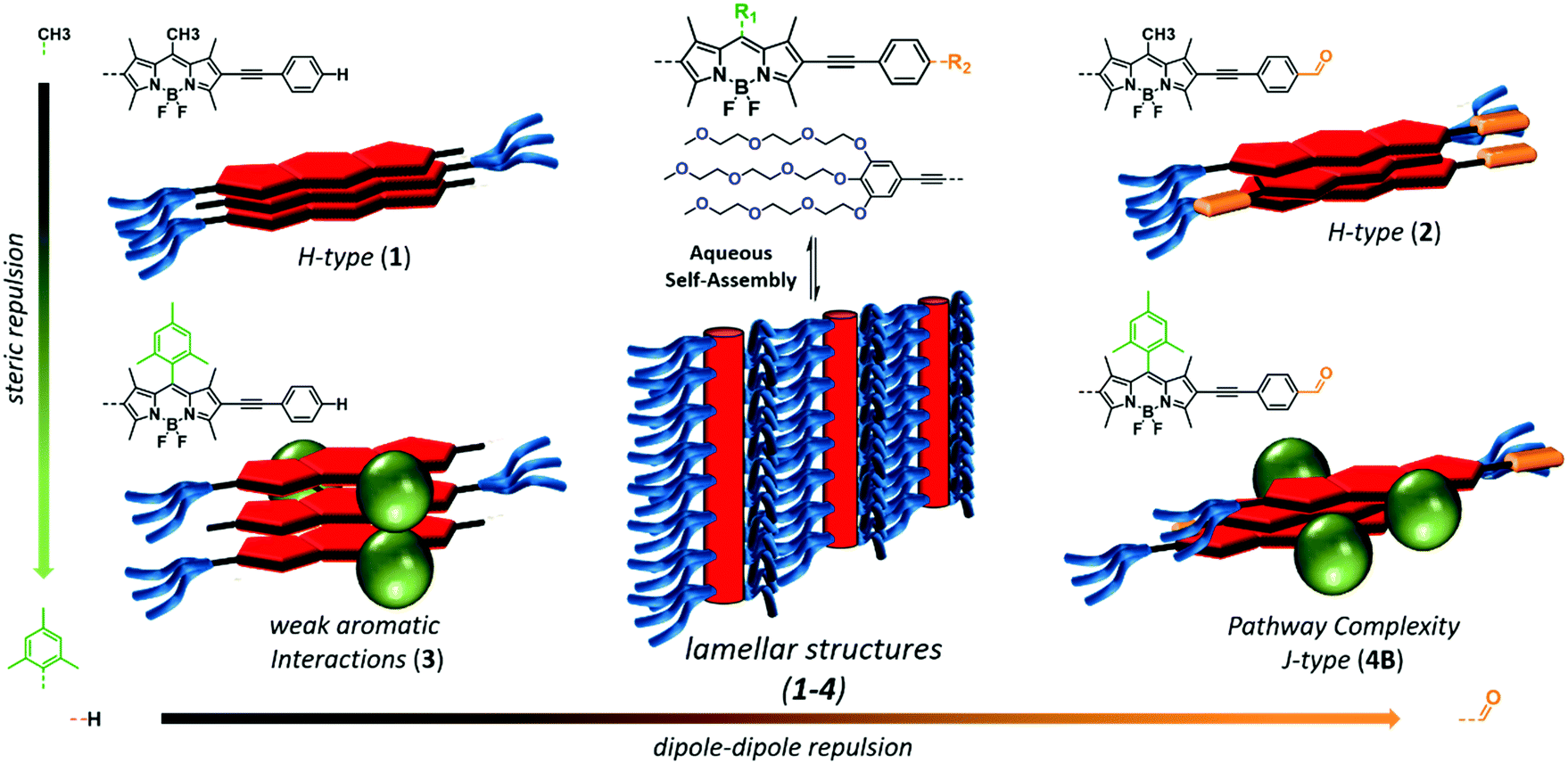
Impact of Positional Isomerism on Pathway Complexity in Aqueous Media
I. Helmers, B. Shen, K. K. Kartha, R. Q. Albuquerque, M. Lee, G. Fernández
Angew. Chem. Int. Ed., 2020, 59,5675-5682
Metallosupramolecular Polymers based on discrete π-conjugated molecules
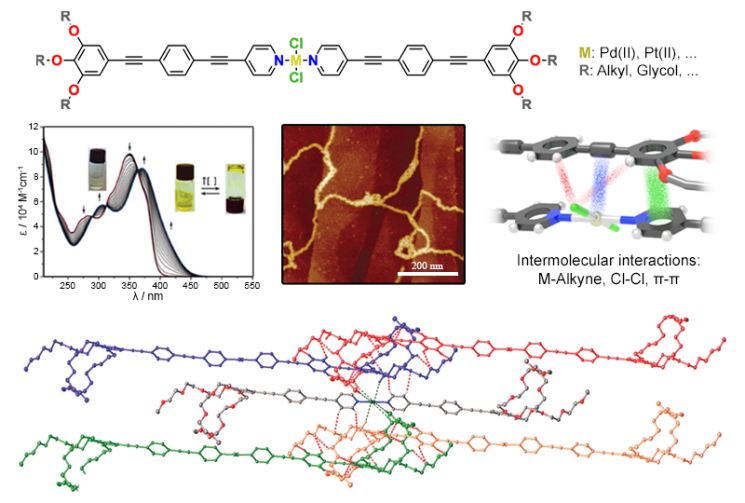
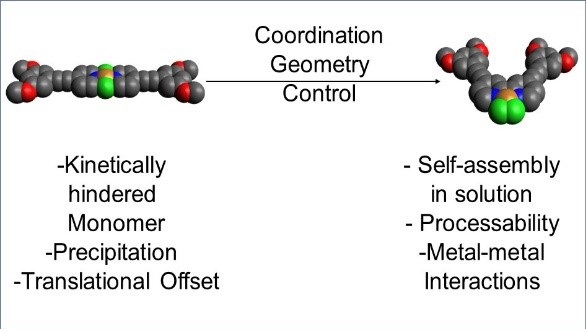
Supramolecular polymers are materials with polymeric properties whose monomer units are held together by reversible non-covalent interactions. One of the most successful strategies to create highly ordered supramolecular polymers consists in introducing aromatic interactions and/or hydrogen bonds. However, the exploitation of other –less conventional- weak forces has been explored to a much lesser extent. In our group, the self-assembly of discrete Pd(II), Zn(II) and Pt(II) metal complexes based on p-conjugated ligands is investigated through a variety of techniques in solution and in the solid state. Due to the presence of multiple polarized atoms, i.e. halogen ligands, metal ions or polarized CH groups, these systems are able to self-assemble into ordered supramolecular polymers driven by multiple weak interactions, such as metal-metal and weak CH hydrogen bonding. The presence of an extended p-surface as well as metal ions make these system potential candidates for optoelectronic applications.
Exploiting coordination geometry to tune the dimensions and processability of metallosupramolecular polymers
N. Bäumer, K. K. Kartha, S. Buss,J. P. Palakkal, C.A. Strassert, G. Fernández
Org. Chem. Front., 2021, 8, 4138
DOI: 10.1039/D1QO00644D
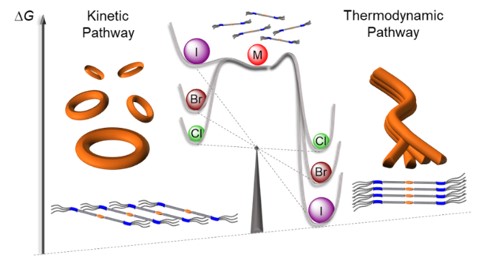
Unraveling Halogen Effects in Supramolecular Polymerization
J. Matern, N. Bäumer, G. Fernández
J. Am. Chem. Soc., 2021, 143, 18
DOI: 10.1021/jacs.1c02384
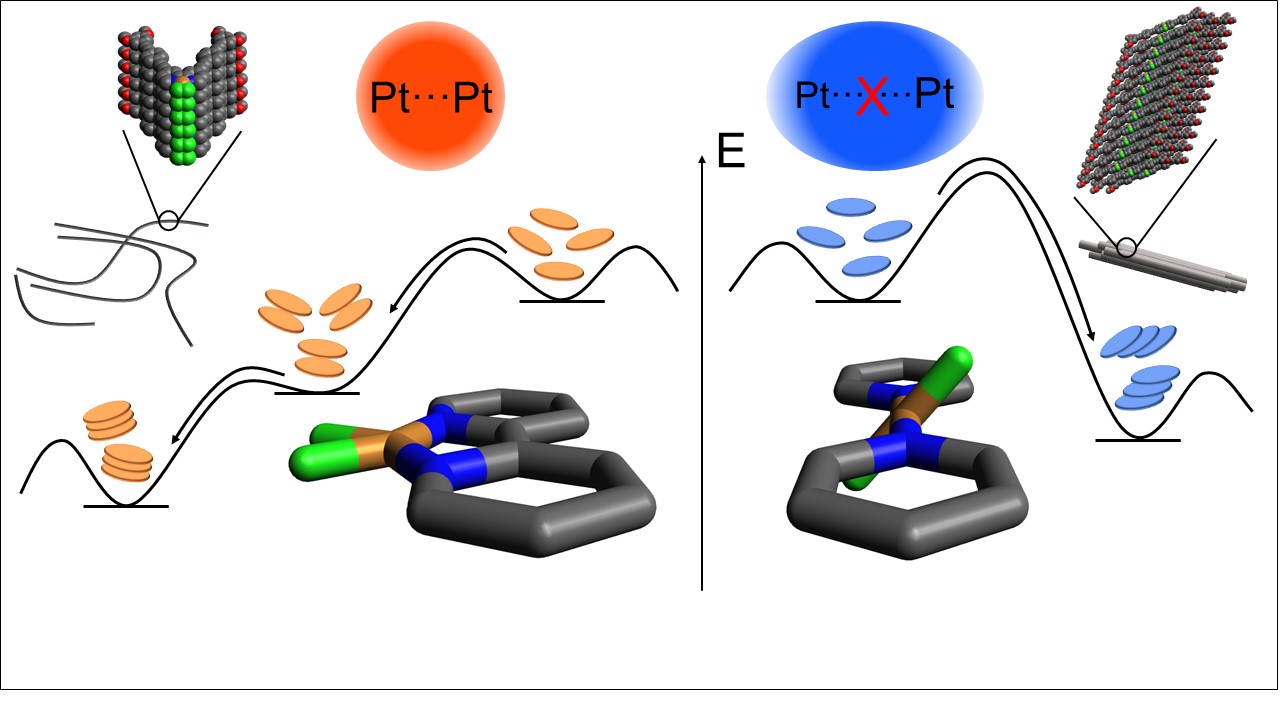
Tuning Energy Landscapes and Metal-Metal Interactions in Supramolecular Polymers regulated by Coordination Geometry
N. Bäumer, K. K. Kalathil, S. Buss, I. Maisuls, J. P. Palakkal, C. A. Strassert, G. Fernandez
Chem. Sci., 2021, 12, 5236
DOI: 10.1039/D1SC00416F
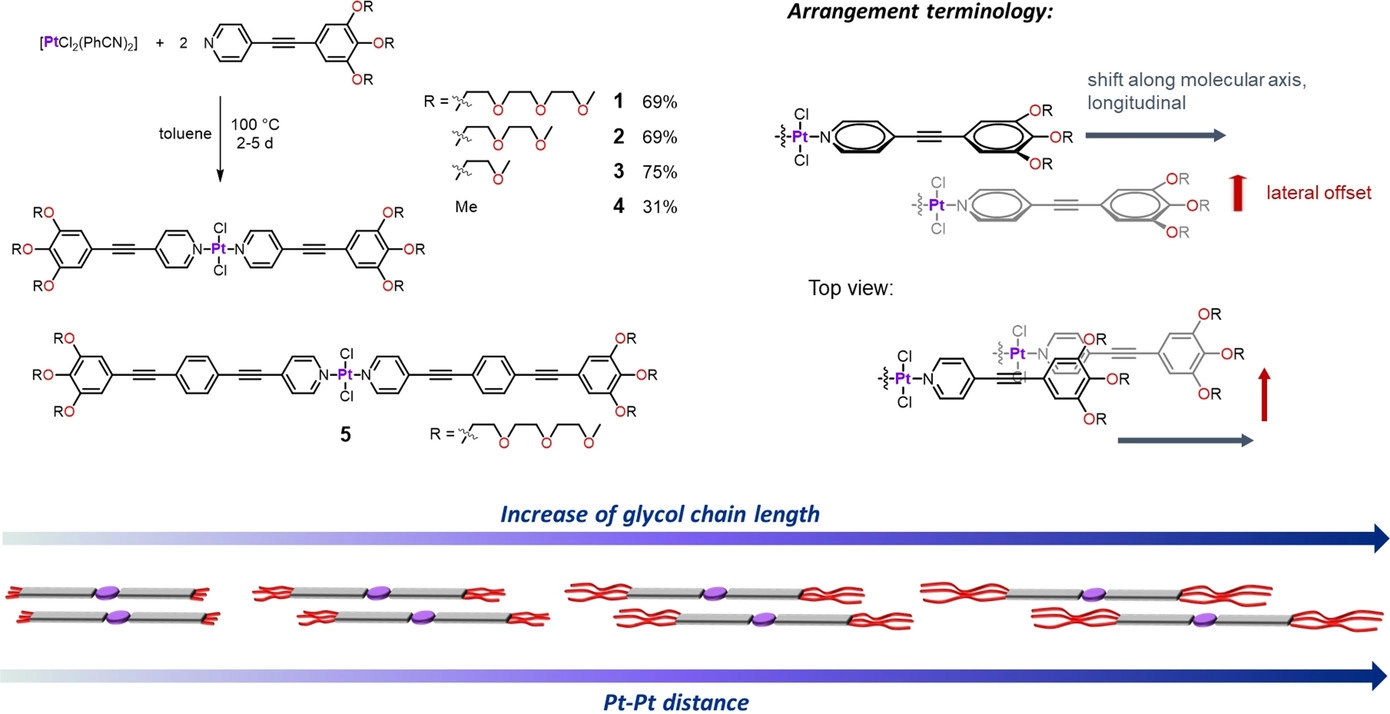
Tuning the Molecular Packing of Self‐Assembled Amphiphilic PtII Complexes by Varying the Hydrophilic Side‐Chain Length
L. Herkert, P. Selter, C. G. Daniliuc, N. Bäumer, J. P. Palakkal, G. Fernández, M. R. Hansen
Chem. Eur. J., 2021, 27,1–11
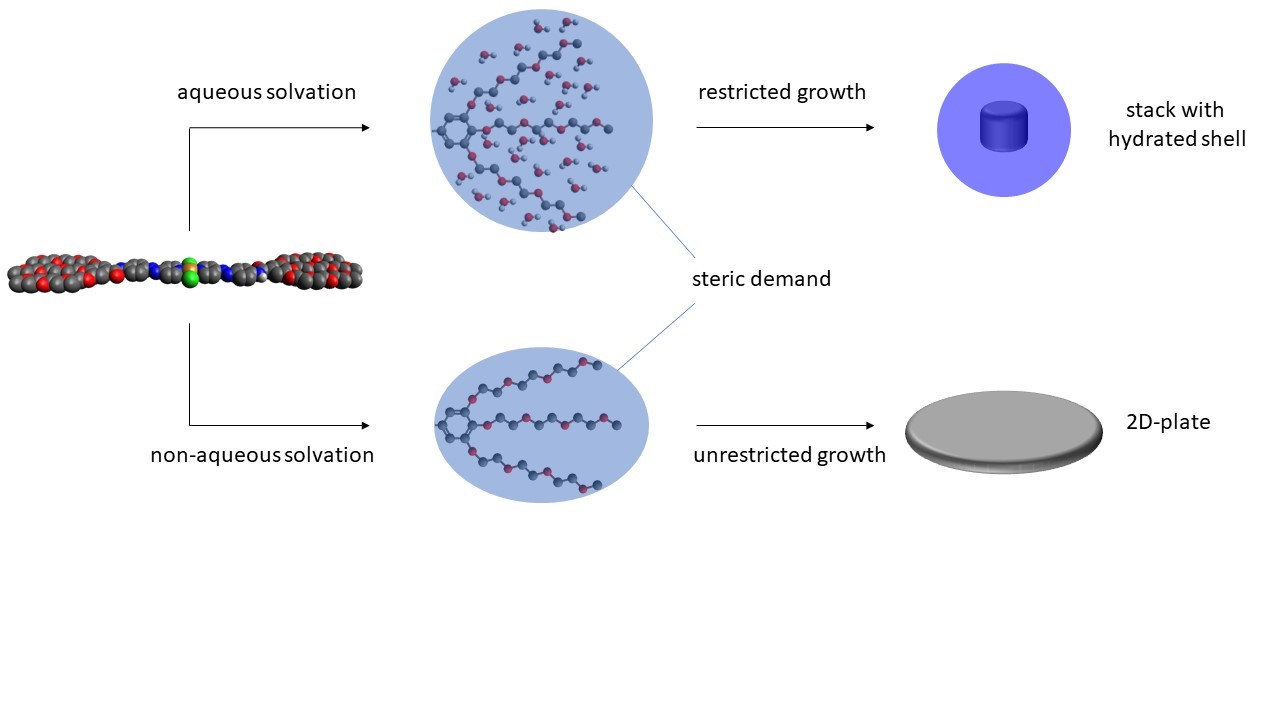
Morphology control in metallosupramolecular assemblies through solvent-induced steric demand
N. Bäumer, K. K. Kartha, J. P. Palakkal, G. Fernández
Soft Matter, 2020,16, 6834-6840
DOI: 10.1039/d0sm00537a
Self-Assembled Structures for Biomedical Applications
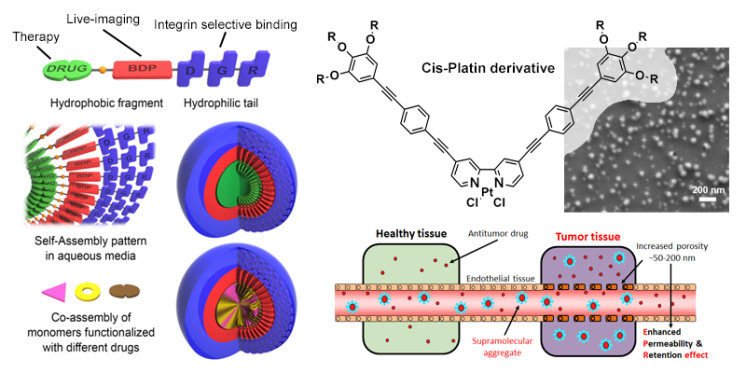
Some of the previously described amphiphilic self-assembled structures have a great potential to be applied in biomedicine, for instance as drug delivery vehicles in cancer therapy. For instance, our group currently tests the ability of various nanoparticle assemblies to encapsulate and transport anticancer drugs by exploiting the enhanced permeability and retention (EPR) effect, by which aggregates of a certain size (usually between 10-200 nm) are prone of accumulation in tumor tissues.
Hydrophobic domain flexibility enables morphology control of amphiphilic systems in aqueous media
I. Helmers, N. Bäumer, G. Fernández
Chem. Commun., 2020, 56, 13808-13811
DOI: 10.1039/D0CC06173E

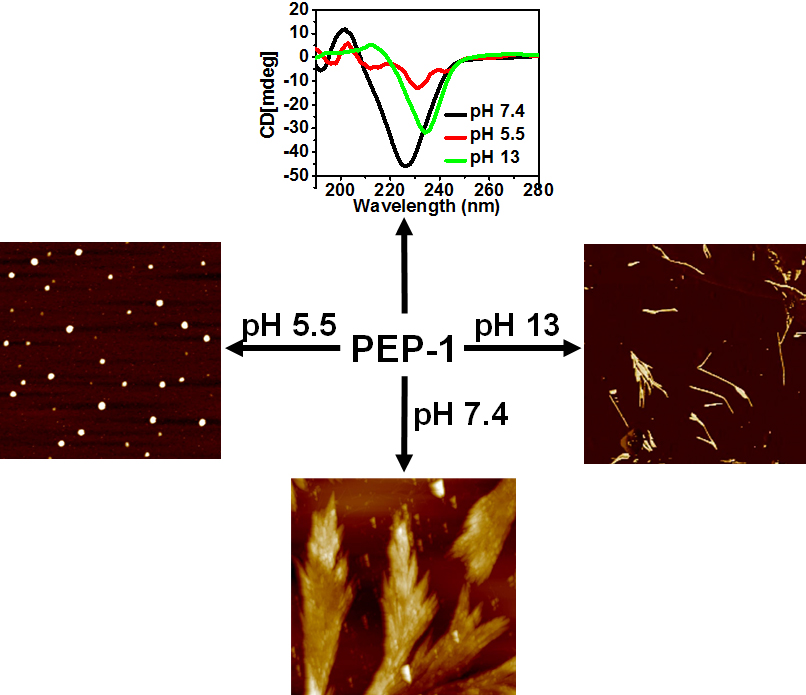
pH- and concentration-dependent supramolecular self-assembly of a naturally occurring octapeptide
G. Ghosh, G. Fernández
Beilstein J. Org. Chem., 2020, 16, 2017–2025
DOI: 10.3762/bjoc.16.168
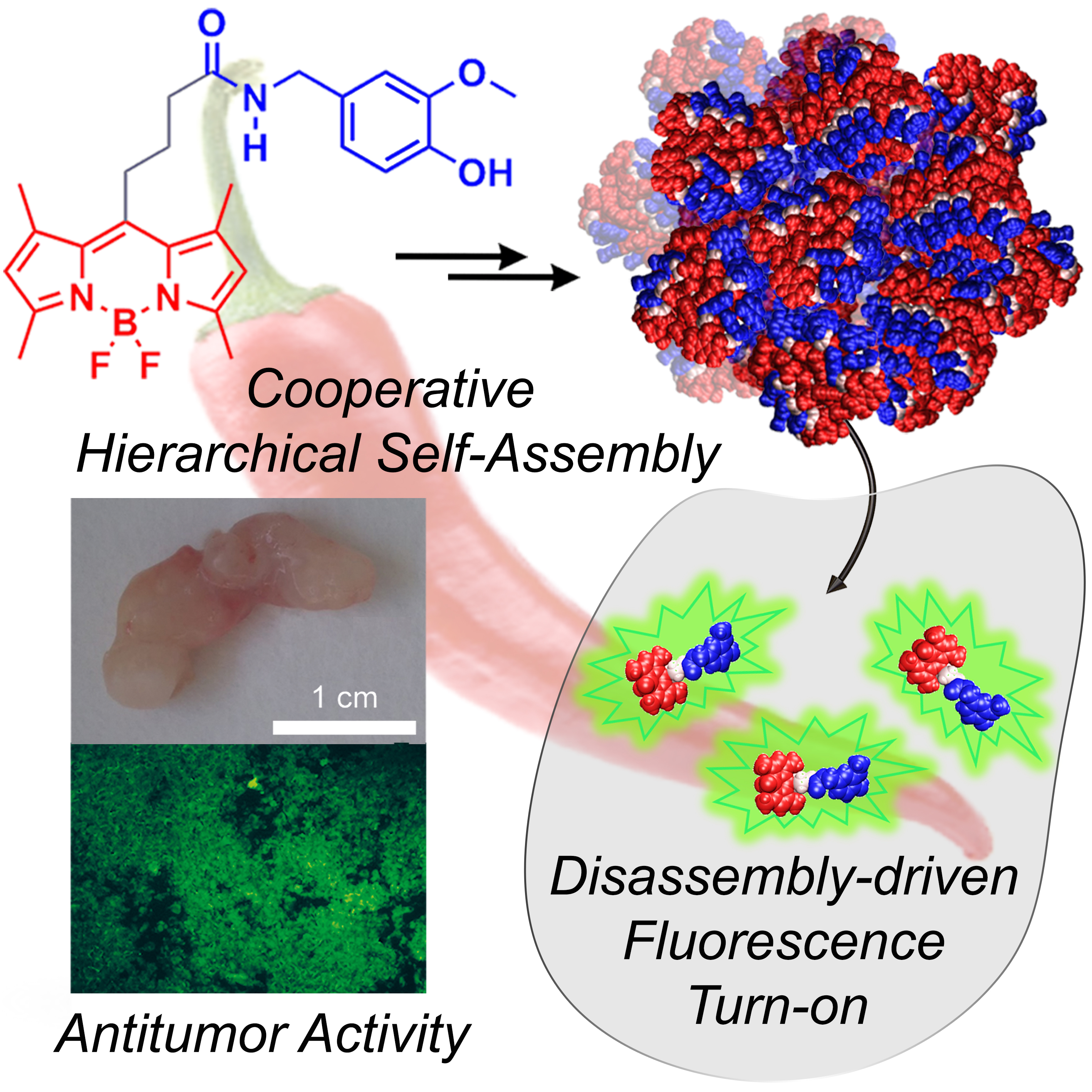
Hierarchical Self-Assembly of BODIPY Dyes as a Tool to Improve the Antitumor Activity of Capsaicin in Prostate Cancer
A. Sampedro, A. Ramos-Torres, C. Schwöppe, C. Mück-Lichtenfeld, I. Helmers, A. Bort, I. Diaz-Laviada Marturet, G. Fernández
Angew. Chem. Int. Ed., 2018, 57, 17235-17239