Blood Vessel Cells on their Travels

The widely branched networks of the blood vessels in the body provide the cells in all its organs with oxygen and nutrients through the blood. They are the transport routes along which immune cells travel to reach whatever place they are needed at in the body, and which hormones use to reach their destination as control signals. It is also via the blood vessels that waste products are transported to the kidney and the liver. This important system is formed at an early stage in the development of the embryo and it reacts flexibly over an entire lifetime – for example, when wounds heal. Biologist and Junior Professor Dr. Wiebke Herzog is looking into the question of how cells in the blood vessels find their way to the right place in the tissue and thus make growth possible. She has now received a Heisenberg fellowship for her work from the German Research Foundation (DFG). The fellowship is initially for three years.
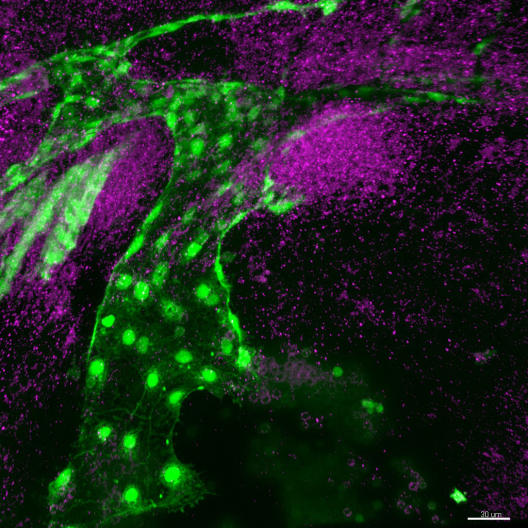
Wiebke Herzog is a member of the Faculty of Biology at Münster University and is a team leader at the Cells-in-Motion Cluster of Excellence. She and her team are housed at the Max Planck Institute for Molecular Biomedicine in Münster. She undertakes her research on the embryos of zebrafish. “Unlike mice or humans, the fish develop in eggs outside the mother’s body,” Wiebke Herzog explains. “During embryonic development, both the embryos and the membranes encasing them are translucent. These are ideal conditions for observing the development of the blood vessels.” The processes which take place in the fish are similar to those in other vertebrates, which is why many of the insights gained are transferable.
Wiebke Herzog is interested specifically in the cells which line the blood vessels on the inside – the so-called endothelial cells. Which molecular signals guide the cells to their place? And how do the cells manage to communicate with one another and travel collectively? In order to find answers to questions such as these, Herzog and her team use modern microscopic methods which enable them to look at living cells as they travel around the organism.
With its prestigious Heisenberg Programme the DFG supports scientists on their way towards a tenured professorship. The aim is for grant-holders to further enhance their scientific reputation in the process.
This article by Christina Heimken was published in the university magazine"wissen|leben" No. 7, 16.11.2016.