Chemists connect three components with new coupling reaction
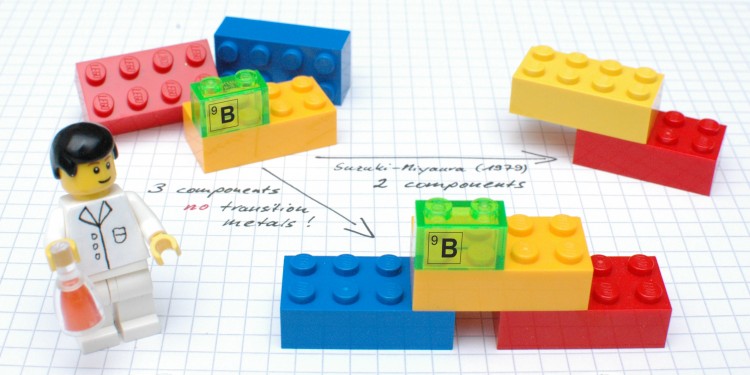
In the current issue of the "Science" magazine, chemists at University of Münster present a new approach which for the first time enables three – and not, as previously, two – chemical components to be "coupled" in one single reaction, without any metals to aid the process. The researchers succeeded in producing not only pharmaceutically relevant compounds containing fluorine, but also various γ-lactones. These organic compounds occur widely in various types of fruit and also, for example, as flavouring substances in whisky and cognac.
"What is remarkable is that for the reaction process no expensive transition metals are needed as catalysts," says Prof. Armido Studer from the Institute of Organic Chemistry at Münster University, the lead author of the study. This represents an important further development of the classic variant, he says, especially with a view to the increasing relevance of sustainable, environmentally-friendly chemistry – so-called green chemistry.
The background: one of the greatest challenges for organic chemists is to create specific bonds between carbon atoms in various chemical components. This is, however, essential for the construction of complex, pharmaceutically active and biologically relevant molecules. "The tools which are particularly important for this are so-called cross-coupling reactions," Studer explains. Probably the most famous example, he adds, is the "Suzuki-Miyaura coupling", which was awarded the Nobel Prize for Chemistry in 2010. This reaction, used by the chemical industry in tonne scale, makes it possible to link two chemical components, although one of the components has to contain a reactive boron moiety. What is decisive for the reaction process, says Studer, is the presence of expensive transition metals such as palladium, which brings the two reactants together, so that in the end a carbon-carbon bond is formed.
The method now developed by the Münster chemists includes the formation of two carbon-carbon bonds. "Unlike classic cross-couplings, however, the valuable boron moiety remains in the product," Studer explains. "At this point, further changes can then be made to the molecules in the same reaction vessel." This method makes it possible, he adds, to produce a large number of different derivatives.
The researchers received financial support from the European Research Council (ERC) for their work.
Original publication:
Marvin Kischkewitz, Kazuhiro Okamoto, Christian Mück-Lichtenfeld, Armido Studer (2017): Radical-polar crossover reactions of vinylboron ate complexes. Science Vol. 355, Issue 6328, pp 936-938; DOI: 10.1126/science.aal3803
