
New method for producing innovative 3D molecules
As its name suggests, ring-shaped “cage molecules” resemble a cage, and it is this three-dimensional structure that makes them significantly more stable than related, flat molecules. Consequently, they could be of interest to drug developers as they represent a possible alternative to conventional molecular rings from the group of aromatic compounds. A research team at the University of Münster led by chemist Prof Dr Frank Glorius has developed a new method for producing so-called heteroatom-substituted 3D molecules and has published the results in the journal "Nature Catalysis". The innovative structures are created by precisely inserting a triatomic unit into the strained (high-energy) ring of a reaction partner.
Aromatic rings are flat rings in organic molecules. They are among the most common ingredients in pharmaceuticals and agrochemicals. However, these structures can be unstable under physiological conditions and thus hamper the effectiveness of pharmaceutical compounds. To solve this problem, scientists have been exploring complex three-dimensional alternatives – cage-like rings that are stiffer and thus more stable. While such 3D substitutes for simple flat rings such as benzene (a ring with six carbon atoms) are already available, it has been much more difficult to synthesise 3D versions of flat rings that contain one or more other important atoms such as nitrogen, oxygen or sulphur. These so-called “heteroaromatic” rings are particularly common in drugs.
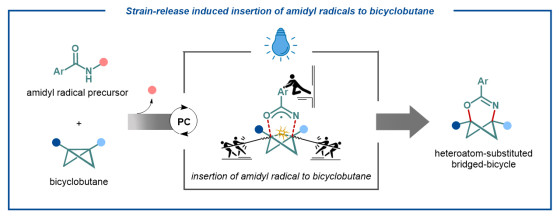
The researchers carried out experimental and computational studies to understand the mechanism of the reaction. They suggest that the reaction is initiated by the light-induced electron transfer event from the excited catalyst to the reactants delivering the final products.
The research project was financed by the German Research Foundation (DFG), the Alexander von Humboldt Foundation and the German Academic Exchange Service.
Original publication
Chetan C. Chintawar, Ranjini Laskar, Debanjan Rana, Felix Schäfer, Nele Van Wyngaerden, Subhabrata Dutta, Constantin G. Daniliuc and Frank Glorius (2024): Photoredox-catalysed amidyl radical insertion to bicyclo[1.1.0]butanes. Nature Catalysis; DOI: 10.1038/s41929-024-01239-9
