AG Bähler - Cell Biology
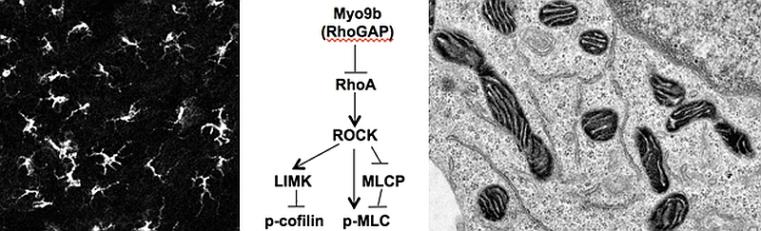

We are studying the functions of molecular motors that exert directed force along actin filaments in mammalian cells. These motor molecules belong to the myosin superfamily. Different myosins show variable motor properties that are well attuned to their cellular functions. We could show that myosin IXB (Myo9b) has an insert in its motor domain that binds the calcium-sensor calmodulin and serves as an actin tether that allows for single-headed processive movement of Myo9b in vitro. In cells, Myo9b motor activity directs recruitment to dynamic actin filaments in extending filopodia and lamellipodia. Myo9b regulates morphology, migration velocity and chemotaxis of leucocytes. It does so by its intrinsic Rho-GTPase-activating protein (RhoGAP) domain that negatively regulates the activity of the small, monomeric G-protein RhoA and associated downstream signaling pathways. The functions of a closely related myosin, myosin IXA (Myo9a), are needed to prevent hydrocephalus formation and kidney disease. A third myosin, myosin XIX (Myo19), is associated with mitochondria and regulates inheritance of mitochondria during cell proliferation.
Experimental strategies comprise the analysis of single molecules to whole organisms and include methods of cell biology, molecular biology, biochemistry, advanced microscopy and mouse technology.
Our goal is to understand in vivo how motors are loading their cargo, choose and use their track(s), identify their destination, stop there and unload their cargo. This information will reveal their precise functions. We will try to answer these and related questions for Myo9b and Myo19, to get a detailed molecular understanding of how Myo9b regulates leukocyte chemotaxis and Myo19 inheritance of mitochondria, respectively. To investigate these questions, we plan to use the most suitable and up to date in vitro and in vivo methods.