Research
Understanding the functions of myosin IX (Myo19)
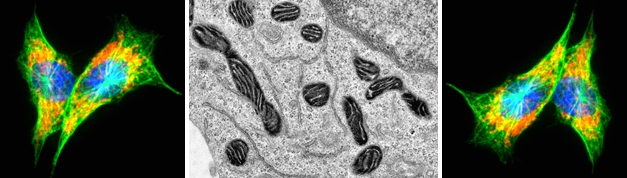
The actin-based motor protein Myo19 associates with mitochondria and couples mitochondrial segregation during mitosis to cell division. Currently, we are elucidating how Myo19 regulates inheritance of mitochondria. To this end we want to identify and characterize mitochondrial receptors for Myo19. Furthermore, we want to analyse how actin-based transport of mitochondria is coordinated with microtubule-based transport during mitosis.
Understanding the functions of myosin IXb (Myo9b)

Directional motility is a fundamental function of immune cells, which are recruited to sites of pathogen invasion or tissue damage by chemoattractant signals. Myosin IXb (Myo9b) is expressed abundantly in immune cells. It contains in its tail region a Rho GTPase-activating protein (GAP) domain. This GAP domain accelerates the inactivation of the monomeric small G-protein Rho by stimulating its GTPase-activity. Thereby, it switches the active GTP-bound form of Rho to the inactive GDP-bound form. Peritoneal macrophages isolated from mice deficient for Myo9b are defective in cell spreading and polarity and are impaired in cell migration and chemotaxis. In these macrophages cell contractility is increased and cell extension driven by actin polymerization is decreased. Thus, the 'motorized Rho inhibitor' Myo9b is a key molecular component required for spatially coordinated cell shape changes and motility.
