Additional Factor for Prolonging Cycle Life of Silicon-based Lithium Ion Batteries Discovered
One approach to further increase the energy density of lithium ion batteries is adding silicon as an active material to the graphite anode. However, volume expansion during charge and discharge cycling causes material stress, which reduces the lifetime of the cells. In contrast, lithium salts, which are the main component of the electrolyte, can extend the lifetime. A team from MEET Battery Research Center, the Institute of Organic Chemistry at the University of Münster, and Helmholtz Institute Münster of Forschungszentrum Jülich, has now investigated in detail the influence of the boron-containing lithium salts lithium bis(oxalato)borate (LiBOB) and lithium difluoro(oxalato)borate (LiDFOB) in silicon-based lithium ion batteries.
Solid Electrolyte Interphase and its Degradation Products Investigated
Initially, the researchers analyzed the solid electrolyte interphase (SEI), the essential passivation layer on the anode that prevents continuous electrolyte decomposition. During their formation, the salts decompose reductively, thus ‘consume’ electrons and deposit as decomposition products on the anode surface. “We discovered that the SEI formed by boron salts are likely more soluble in the electrolyte and can diffuse to the cathode,” says MEET scientist Dr Anindityo Arifiadi. In this ‘crosstalk’ of the SEI decomposition products, the research team identified another important factor affecting cycle life caused by the use of LiBOB or LiDFOB. “When these products reach the cathode, they get oxidized, and the apparently lost electrons of the reduced species can be returned to the cell and this way recover the apparently lost capacity,” explains Arifiadi.
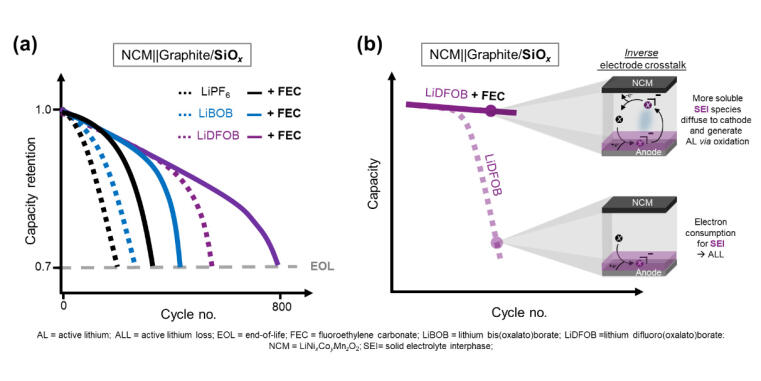
Further research and development are now required for the commercial application of boron-containing lithium salts. Dr Johannes Kasnatscheew, Head of the Research Division Materials at MEET Battery Research Center, evaluates: "LiBOB and LiDFOB still gas too much to be used in practice. However, our findings on their effectiveness in terms of capacity recovery are an important step toward further optimizing electrolytes for lithium ion batteries with silicon anodes."
Entire Study Available
The detailed results have been published by the authors Dr Anindityo Arifiadi, Dr Jaroslav Minar, Linus Voigt, Dr Dominik Voigt, Marc Vahnstiege, Julius Buchmann, Dr Feleke Demelash, Dr Simon Wiemers-Meyer, Dr Sascha Nowak and Dr Johannes Kasnatscheew, MEET Battery Research Center, Ankita Das und Prof. Dr Frank Glorius, Institute of Organic Chemistry at the University of Münster, Dr Peng Yan, Helmholtz Institute Münster, as well as Prof. Dr Martin Winter, MEET Battery Research Center and Helmholtz Institute Münster, in the journal “Small Science”.

