PROJECT A08
Molecular Parameters for the Dynamic Assembly of Hedgehog Signaling Platforms
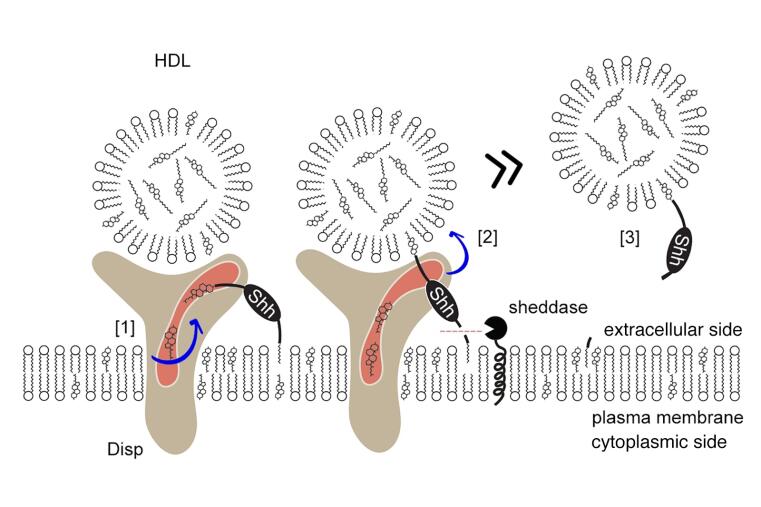
The regulated solubilization of dual-lipidated (N-terminally palmitoylated, C-terminally cholesteroylated) Sonic hedgehog (Shh) is one way to activate the Shh signaling pathway in neighboring cells. Solubilization starts at the plasma membrane (PM) of sender cells and involves the 12-pass transmembrane protein Dispatched (Disp). In the previous funding period, we showed that Disp specifically extracts the cholesterol-modified C-terminal Shh peptide from the PM and transfers it to soluble high-density lipoprotein (HDL) acceptors, and that the transfer is completed by proteolytic processing of the palmitoylated N-terminal membrane anchor of Shh. The resulting HDL-associated Shh is highly bioactive. We now aim to characterize the following step in Shh signaling: the mechanistic mode by which HDL-associated Shh forms biological gradients that are robust yet scalable at cellular interfaces.
While our data explain how Disp solubilizes cholesteroylated Shh from the PM, we also observed that Disp transfers free (non-esterified) PM cholesterol to HDL. As a consequence, Disp-deficient cells accumulate free cholesterol and strongly downregulate the expression of genes involved in cholesterol uptake or biosynthesis. This newly discovered non-canonical function of Disp as a major exporter of free PM cholesterol affects multiple aspects of cellular behavior: Our preliminary transcriptomic data indicate that the expression of several gene clusters related to cell adhesion and migration is affected in Disp-deficient cells, likely as a consequence of persistently elevated PM cholesterol levels. We thus also analyze how Disp affects cellular physiology independently of its well-established role in regulating Shh signaling.


