AG Grashoff - Quantitative Cell Biology
Scientific Background
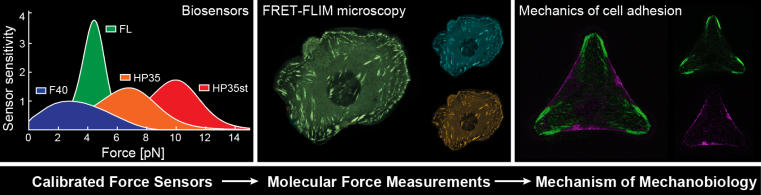
The ability of cells to sense and respond to mechanical forces is essential for all known organisms. It has been recognized that cellular mechanosensitivity is crucial in humans too and plays a crucial role during organ development and tissue maintenance. It is also clear that the inability to properly process mechanical signals can trigger disease development. How mechanical signals are processed in our cells, however, is largely unknown. One reason for the inability to understand molecular processes of force transduction is that forces acting across individual molecules in cells are very small – in the order of just a few piconewton (pN) – which is technically difficult to measure. As a result, it was unknown for many decades, when and where distinct proteins experience mechanical forces inside cells. Therefore, we have developed a microscopy-based technique to quantify and visualize pN-scale forces in cells (Grashoff et al, Austen et al, Ringer et al, Fischer et al) . We utilize and continuously develop this, by now world-wide applied, technology to explore how mechanical signals are born and propagate intracellularly. We complement our experiments with single-molecule super-resolution microscopy (Fischer et al, Fischer et al), and we develop novel optogenetic tools to modulate molecular linkages in cells with light. Together, these experiments allow us to define the molecular mechanisms underlying force transduction and thereby develop a better understanding of various mechanobiological processes in cells. Most of our experiments are based on mammalian cell systems and we use genetic mouse models to test physiological relevance of our observations (Kanoldt et al).
