Daphnia is a model organism for ecological and physiological research.
1. Oxygen transport in Daphnia at variable oxygen conditions
- Optophysiological analyses of the oxygen transport cascade in Daphnia
- Structure and function of Daphnia hemoglobins (Hb): Hb genes & synthesis sites, Hb induction (transcription factors, Hb mRNA & subunits), subunit composition, heterogeneity and quarternary structure of Hb macromolecules
- Adjustments to variable oxygen conditions on the behavioural level (oxygen preference)
- Adjustments to variable oxygen conditions on the biochemical level (proteome, protein structure and function, metabolites)
Selected references:
Paul, Zeis, Lamkemeyer, Seidl, Pirow (2004) Control of oxygen transport in the microcrustacean Daphnia: regulation of haemoglobin expression as central mechanism of adaptation to different oxygen and temperature condtions. Acta Physiol Scand 182: 259-275
Lamkemeyer, Paul, Stöcker, Yiallouros, Zeis (2005) Macromolecular isoforms of Daphnia magna haemoglobin. Biol Chem 386: 1087-1096
Lamkemeyer, Zeis, Decker, Jaenicke, Waschbüsch, Gebauer, Markl, Meissner, Rousselot, Zal, Paul (2006) Molecular mass of macromolecules and subunits and quarternary structure of haemoglobin from the microcrustacean Daphnia magna. FEBS J 273: 3393-3410
Zeis, Lamkemeyer, Paul, Nunes, Schwerin, Koch, Schütz, Madlung, Fladrer, Pirow (2009) Acclimatory responses of the Daphnia pulex proteome to environmental changes. I. Chronic exposure to hypoxia affects the oxygen transport system and carbohydrate metabolism. BMC Physiol 9: 7
Weber, Pirow (2009) Physiological responses of Daphnia pulex to acid stress. BMC Physiol 9 :9
Schwerin, Zeis, Horn W, Horn H, and Paul (2010) Hemoglobin concentration in Daphnia (D. galeata-hyalina) from the epilimnion is related to the state of nutrition and the degree of protein homeostasis. Limnol Oceanogr 55: 639–652
Gerke P, Börding C, Zeis B, Paul RJ (2011) Adaptive haemoglobin gene control in Daphnia pulex at different oxygen and temperature conditions. Comp Biochem Physiol A 159:56-65
Zeis B, Becker D, Gerke P, Koch M, Paul RJ (2013) Hypoxia-inducible haemoglobins of Daphnia pulex and their role in the response to acute and chronic temperature increase. BBA Proteomics 1834:1704-1710
Cuenca Cambronero M, Zeis B, Orsini L (2017) Haemoglobin-mediated response to hyper-thermal stress in the keystone species Daphnia magna. Evol Appl 11:112-120
2. Thermal acclimation and adaptation in Daphnia

- Thermal adjustments in Daphnia on the physiological and behavioural level (e.g. oxygen transport and energy provision, thermal preference, activity patterns)
- Thermal adjustments in Daphnia on the biochemical level (e.g. proteome, protein structure and function, metabolites)
- Relationship between seasonal changes of abiotic and biotic environmental factors, the clonal structure of Daphnia populations, and the clone-specific physiology and biochemistry of Daphnia including acclimatory adjustments and capacities
- Laboratory selection experiments on Daphnia
Selected references:
Lamkemeyer, Zeis, Paul (2003) Temperature acclimation influences temperature-related behaviour as well as oxygen-transport physiology and biochemistry in the water flea Daphnia magna. Can J Zool 81: 237-249
Paul, Lamkemeyer, Maurer, Pinkhaus, Pirow, Seidl, Zeis (2004) Thermal acclimtion in the microcrustacean Daphnia: a survey of behavioural, physiological and biochemical mechanisms. J Therm Biol 79: 655-662
Seidl, Pirow, Paul (2005) Acclimation of the microcrustacean Daphnia magna to warm temperatures is dependent on haemoglobin expression. J Therm Biol 30: 532-544
Pinkhaus, Schwerin, Pirow, Zeis, Buchen, Gigengack, Koch, Horn, Paul (2007) Temporal environmental change, clonal physiology and the genetic structure of a Daphnia assemblage (D. galeata-hyalina hybrid species complex). Freshwater Biol 52: 1537-1554
Schwerin, Zeis, Lamkemeyer, Paul, Koch, Madlung, Fladerer, Pirow (2009) Acclimatory responses of the Daphnia pulex proteome to environmental changes. II. Chronic exposure to different temperatures (10 and 20 °C) mainly affects protein metabolism. BMC Physiol 9: 8
Zeis, Horn, Gigengack, Koch, Paul (2010) A major shift in Daphnia genetic structure after the first ice-free winter in a German reservoir. Freshwater Biology 55: 2296-2304
Hülsmann S, Wagner A, Pitsch M, Horn W, Paul R, Rother A, Zeis B (2012) Effects of winter conditions on Daphnia dynamics and genetic composition in a dimictic temperate reservoir. Freshwater Biology 57: 1458-1470
Paul RJ, Mertenskötter A, Pinkhaus O, Pirow R, Gigengack U, Buchen I, Koch M, Horn W, Zeis B (2012) Seasonal and interannual changes in water temperature affect the genetic structure of a Daphnia assemblage (D. longispina complex) through genotype-specific thermal tolerances Limnol. Oceanogr., 57:619-633
3. Stress physiology of Daphnia
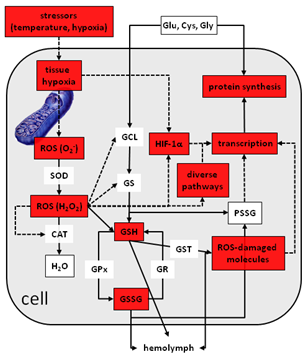
- ROS and redox signals as well as signals related to macromolecular damage are key regulators of cellular stress responses. Acute temperature and oxygen stress trigger distinctive oscillatory ROS and glutathione redox signals, which are linked to both stress intensity (tissue hypoxia) and stress response (gene expression). Time-resolved measurements for detecting temporal patterns turned out to be a good strategy to assess these stressor-signal-effect relationshipsin two Daphnia species (D. pulex and D. magna), for which differences in system dynamics have been proven.
- Analyses focus on:
- 1. Connections between stressors, stress signals, transcription factor levels, and stress responses upon temperature and oxygen stress
- 2. Gene expression patterns under stress, and to understand the molecular principles and control of CSR (instantaneous stress defense) and CHR (acclimation)
a) the application of temperature and oxygen stress,
b) the determination of stress signals (ROS level, glutathione redox state, macromolecular damage),
c) the measuring of stress-related transcription factor levels,
d) the evaluation of stress responses by assessing gene expression on the transcript (real-time RT-PCR, microarray analysis) and protein (Western blot, enzymatic assay, proteomics) levels,
d) the analysis for common stress-related transcription factor binding motifs (e.g., HRE, ARE, TRE) in genes similarly regulated in time, and
e) the verification of model concepts e.g., by applying modulated test signals (ROS, glutathione) and measuring transcription factor levels and gene expressions. - 3. effects of starvation as stressor on stress signals and stress responses with the main focus on iron metabolism and homeostasis
- 4. relationship between de novo glutathione and protein synthesis, effects of elevated protein synthesis rate on antioxidant buffering capacity
Selected references:
Becker D, Brinkmann B, Zeis B, Paul R (2011) Acute changes in temperature or oxygen availability induce ROS fluctuations in Daphnia magna linked with fluctuations of reduced and oxidized glutathione, catalase activity and gene (haemoglobin) expression. Biology of the Cell 103:351-363
