Nanobodies and antibody-like proteins as tools in diagnostics and controllable protein-protein interactions
Nanobodies are single-domain antibody fragments derived from heavy-chain-only antibodies. Due to their small size, easier expression in bacterial cells and high stability, compared to regular IgG antibodies, they are attractive for many applications in diagnostics, therapy and basic research.
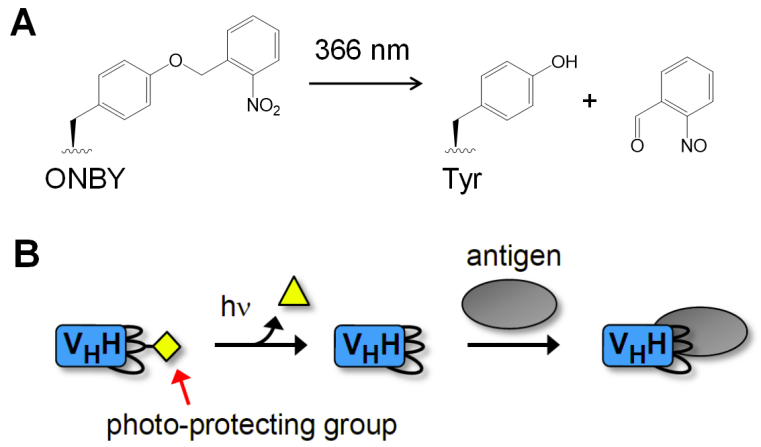
We and others[1] have designed the first nanobodies that are blocked in binding their antigen until they are activated by irradiation with light. The concept is that a critical tyrosine side chain in the paratope region of the nanobody is substituted by the unnatural amino acid (ncAA) ortho-nitrobenzyl tyrosine (ONBY; Figure 1). Removal of the photolabile protection group reconstitutes the tyrosine side chain and hence the native nanobody. Incorporation of the ncAA is performed by expressing of the protein in E. coli using genetic code expansion.[2] We have shown for several nanobodies that this design concept allows generation of so-called photobodies with changes in dissociation constants of >1,000 to 10,000-fold to control protein interactions on demand within seconds.[3-4] Furthermore, we have transduced these photo-caged nanobodies inside mammalian cells to manipulate endogenous cellular processes with the high spatiotemporal resolution of light.[5]
References
[1] T. Bridge, S. A. Shaikh, P. Thomas, J. Botta, P. J. McCormick, A. Sachdeva, Angew Chem Int Ed Engl, 2019, 58, 17986-17993. DOI
[2] A. Deiters, D. Groff, Y. Ryu, J. Xie, P. G. Schultz, Angew Chem Int Ed Engl, 2006, 45, 2728-2731. DOI
[3] B. Jedlitzke, Z. Yilmaz, W. Dörner, H. D. Mootz, Angew Chem Int Ed Engl, 2020, 59, 1506-1510. DOI
[4] B. Jedlitzke, H. D. Mootz, Chembiochem, 2022, 23, e202200079. DOI
[5] B. Jedlitzke, H. D. Mootz, ChemPhotoChem, 2021, 5, 22-25. DOI