Lipid droplets
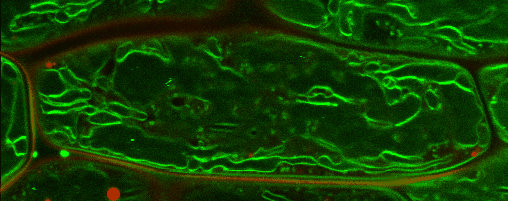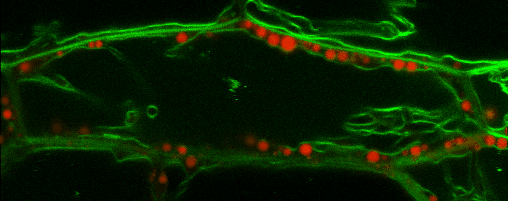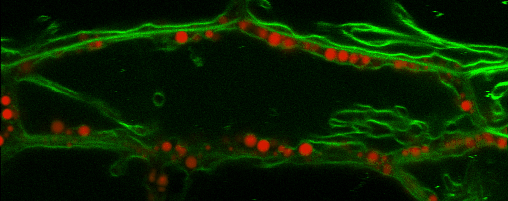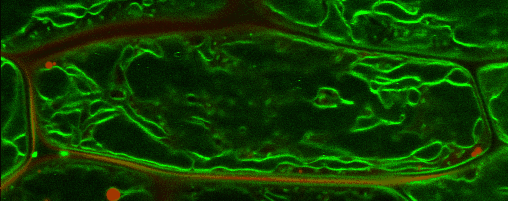
Lipid droplets (LDs) are special organelles consisting of a phospholipid monolayer surrounding a core of hydrophobic compounds, predominantly triacylglycerol (TAG). They are very prominent in seeds of angiosperms or the tapetum and the cytosol of pollen grains, but principally occur in all plant tissues in varying numbers.
For many years, LDs were regarded to be mere carbon storage organelles that provide energy (e.g. for heterotrophic growth stages like seed germination and seedling establishment). In the last years, however, it became more and more evident that these underrated organelles take an active part in many cellular and physiological processes as for example during abiotic and biotic stress. Yet, numerous questions about LD biology and their cellular roles remain to be resolved. One reason for this poor understanding of LDs is the small number of proteins actually known to reside there. While hundreds of potential candidates have been found in proteomic studies of lipid droplets from higher plants and algae, these were to the largest part never validated using a second line of evidence.
In our group, we therefore combine quantitative proteomics of LDs and specialized cell biological techniques developed in our lab to identify and verify potential candidates. In addition, the functional characterization of these identified proteins poses a big part of our research. For this, we make use of different genetic tools (e.g. CRISPR/Cas9) as well as biochemical (LC/MS-based proteomics), bioanalytical (different mass spectrometric analyses) and cell biological approaches (e.g. confocal laser scanning microscopy).
Due to their inert nature and capacity to store large amounts of hydrophobic compounds, LDs are optimally suited as factories and storage sites for valuable metabolites or defense compounds.
We want to exploit this in biotechnological approaches.