Research concept
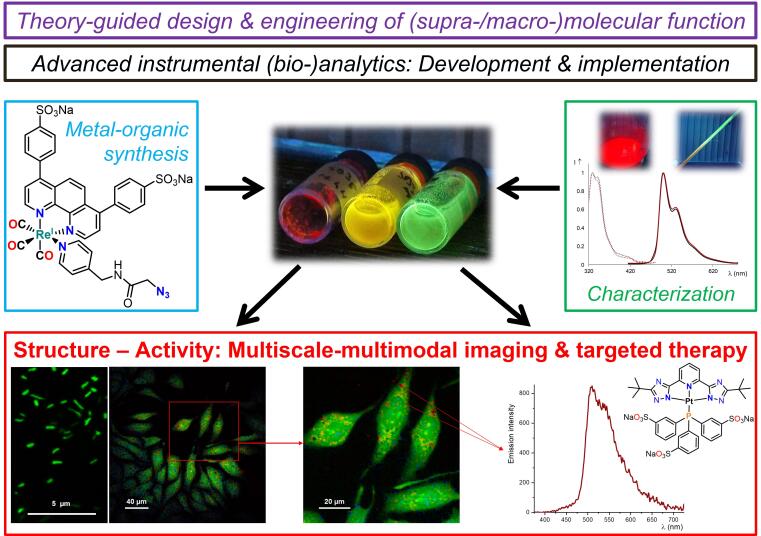
The Strassert Lab is devoted to theory-guided design and metal-organic synthesis of molecular entities. It encompasses organic compounds with main-group elements as well as functional ligands for d- and f-block cations across the periodic table, while exploring diverse binding modalities, preparative strategies, redox properties, relativistic and spin effects.
By custom-tailoring computational tools and instrumental analysis, coordination compounds are engineered for optical manipulation of structural features and dynamic processes in biomacromolecules or supramolecular assemblies as well as in living systems towards tailored probes for multiscale-multimodal bioimaging, thereby acting as contrast agents or luminescent markers. Consequently, spectroelectrochemical techniques and time-resolved multiphoton microspectroscopic methods are developed to apply these new entities to high-resolution optical microscopy.
Another goal is MRI, photoacoustic, or radioactive labeling, along with the targeted ROS-driven inactivation of antibiotic-resistant pathogens and neoplastic cells by implementing custom-made NIR absorbers for in vivo therapy and diagnostics. Research efforts on molecular photosensitizers and emitters are also oriented towards printable devices employing soft architectures and solution-processable fluorophores or phosphors by investigating their coupling with (semi)conductive surfaces for oxygen sensing or electrochemicaluminescence.
The quantitative understanding of macromolecular function and structure-property-activity relationships is also relevant for optobioelectronics as well as in photoinduced charge separation processes. This knowledge will also be useful for applications in photo(bio)catalysis or energy conversion, as well as for the integration of hybrid devices.
Selected publications co-authored by Prof. C. A. Strassert (related to the research concept)
60. "A new phosphorus-rhodamine voltage-sensing dye for optical membrane potential imaging." Höhl, M. et al., Photochemical and Photobiological Sciences 2026, DOI: 10.1007/s43630-026-00903-2
59. "Covalent Attachment of a Platinum(II) Complex inside DNA Duplexes". Schäfer, T. et al., Chemistry Europe 2025, DOI: 10.1002/ceur.202500416
58. "Host−Guest Nanoparticles Incorporating a Fluorophore-Conjugated Pt(II) Complex: A Ratiometric Dual Emitter Performing as a Self-Referenced Oxygen Reporter." Rex, T. et al., ACS Applied Optical Materials 2026, DOI: 10.1021/acsaom.5c00262
57. "Steric Pressure in Heteropentacenes Modulates the Photophysical Properties – A Molecular Design Strategy for Functional Materials." Huber, A. et al., Chemical Science 2025, 16, 15723
56. "Monte Carlo simulations of transverse relaxation for characterization of physicochemical properties of superparamagnetic iron oxide nanoparticles." Klünder, L. et al., Magnetic Resonance Imaging 2026, 125, 10.1016/j.mri.2025.110553
55. "Visualization of KCa3.1 Channels in Tumor Cells by Optimized Senicapoc-Bodipy Conjugates." Thale, I. et al., ACS Pharmacology & Translational Science 2025, 8, 3371
54. "Covalently Platinated DNA Oligonucleotides as Ratiometric Dioxygen Sensors." Schäfer, T. et al., Bioconjugate Chemistry 2025, 36, 2487
53. "Controlling Asymmetry Amplification through Supramolecular Polymorphism." Montañez-Moyano, C. et al., Angewandte Chemie International Edition 2025, 64, 10.1002/anie.202509031
52. "Photodynamic ROS inducers delivered via electrostatic antibody targeted (ELART) nanocarriers incorporate into tumour cells and inhibit colony growth." Faust, A. et al., Chemical Communications 2025, 61, 8079
51. "Heterobimetallic contacts in statistical co-crystals of homoleptic coordination compounds with ligand-encoded H-F bonds: structure, photophysics and mechano-responsive properties." Theiss, T. et al., Chemical Communications 2025, 61, 1387
50. "Supramolecular Assembly of Water-Soluble Platinum(II) Complexes: From Emission Modulation to Cell Imaging in Specific Organelles." Rex, T. et al., Chemistry - A European Journal 2025, 31, 10.1002/chem.202404432
49. "Organic Room-Temperature near-IR Phosphorescence Harvested by Intramolecular Through-Space Sensitization in Composite Molecules." Partanen, I. et al., Angewandte Chemie International Edition 2025, 64, 10.1002/anie.202503327
48. "Aggregation, Photoluminescence, and Cytotoxicity of Pt(II) and Re(I) Complexes Bearing Multimodal-Coordinating Luminophores." Buss, S. et al., Chemistry - A European Journal 2025, 31, 10.1002/chem.202404115
47. "Fluorescent Probes to Image the KCa3.1 Channel in Tumor Cells." Thale, I. et al., Pharmaceutics 2025, 17, 10.3390/pharmaceutics17020154
46. "Reversible Piezochromism of Platinum(II) and Palladium(II) Dimers in Molecular Single Crystals." Steeger, P. et al., Nano Letters 2025, 25, 2628
45. "Water-soluble luminescent platinum(II) complexes for guanine quadruplex binding." Kroos, S et al., Dalton Transactions 2025, 54, 5367
44. "In situ Cyclization of Aromatic Thioethers in Emissive Materials to Generate Phosphorescent Dibenzothiophenes." Schmiedtchen, M. et al., Angewandte Chemie International Edition 2025, 64, 10.1002/anie.202414326
43. "Molecular folding governs switchable singlet oxygen photoproduction in porphyrin-decorated bistable rotaxanes." Riebe, J. et al., Communications Chemistry 2024, 7, 10.1038/s42004-024-01247-7
42. "Merging of a Supramolecular Ligand with a Switchable Luminophore - Light-Responsiveness, Photophysics and Bioimaging." Balszuweit, J. et al., Chemistry - A European Journal 2024, 30, 10.1002/chem.202402578
41. "Adaptive photoluminescence through a bioinspired antioxidative mechanism." Rex, T. et al., Chemical Science 2024, 15, 18881
40. "Antiprotozoal Pt(II) Complexes as Luminophores Bearing Monodentate P/As/Sb-Based Donors: An X‑ray Diffractometric, Photoluminescence, and 121Sb-Mössbauer Spectroscopic Study with TD-DFT-Guided Interpretation and Predictive Extrapolation toward Bi." Buss, S. et al., Inorganic Chemistry 2024, 63, 10114
39. "Functional Pt(II) and Re(I) Complexes with CO- and β-Carboline-Based Coligands: From Time-Resolved Photoluminescence Spectroscopy and Evaluation of 1O2 Photosensitization Efficiency Toward in vitro (Photo)cytotoxicity." Kirse, T. M. et al., Organometallics 2024, 43, 1752
38. "Assessing the Character of the C6F5 Ligand from the Electrochemical and Photophysical Properties of [Ni(C6F5)2(N^N)] Complexes." Jordan, R. et al., Inorganic Chemistry 2024, 63, 11079
37. "Activating the Fluorescence of a Ni(II) Complex by Energy Transfer." Hung, T.-C. et al., Journal of the American Chemical Society 2024, 146, 8858
36. "Cyanido-bridged diplatinum(II) complexes: ligand and solvent effect on aggregation and luminescence." Khistiaeva, V.V. et al., Chemical Science 2024, 15, 4005 35. On the thermodynamics of aggregation toward phosphorescent metallomesogens: From electronic tuning to supramolecular design. Gutierrez Suburu, M. E. et al., Aggregate 2024, 5, 10.1002/agt2.473
34. "Superparamagnetic Nanoparticles with Phosphorescent Complexes as Hybrid Contrast Agents: Integration of MRI and PLIM." Rivas Aiello, M. B. et al., Small Science 2024, 4, 10.1002/smsc.202300145
33. "One Dianionic Luminophore with Three Coordination Modes Binding Four Different Metals: Toward Unexpectedly Phosphorescent Transition Metal Complexes." Kirse, T. et al., Advanced Science 2023, 11, 10.1002/advs.202306801
32. "Access to Strong Thieno[3,2-b]phosphole-Based Solid-State Emitters via Manganese(III)-Mediated Oxidative Annulation." König, N. et al., Chemistry of Materials 2023, 35, 8218
31. "Room-temperature phosphorescence from Pd(II) and Pt(II) complexes as supramolecular luminophores: The role of self-assembly, metal-metal interactions, spin-orbit coupling and ligand-field splitting." Theiss, T. et al., Journal of the American Chemical Society 2023, 145, 3937
30. "Site-specific covalent metalation of DNA oligonucleotides with phosphorescent platinum(II) complexes." Boisten, F. et al., Chemical Science 2023, 14, 2399
29. "Room-temperature phosphorescence from Pd(II) and Pt(II) complexes as supramolecular luminophores: The role of self-assembly, metal-metal interactions, spin-orbit coupling and ligand-field splitting." Theiss, T. et al., Journal of the American Chemical Society 2023, 145, 3937
28. "Facile modification of phosphole-based aggregation-induced emission luminogens with sulfonyl isocyanates." König, N. et al., Chemical Science 2023, 14, 2267
27. "Electrostatic anti‑CD33‑antibody–protamine nanocarriers as platform for a targeted treatment of acute myeloid leukemia." Bäumer, N. et al., Journal of Hematology & Oncology 2022, 15, 171
26. "Genetically encoded dual fluorophore reporters for graded oxygen-sensing in light microscopy." Bauer, N. et al., Biosensors and Bioelectronics 2023, 221, 114917
25. "Luminescence and Length Control in Nonchelated d8-Metallosupramolecular Polymers through Metal-Metal Interactions." Matern, J. et al., Angewandte Chemie International Edition 2022, 61, e202208436
24. "Monoanionic C^N^N Luminophores and Monodentate C-Donor Co-Ligands for Phosphorescent Pt(II) Complexes: A Case Study Involving Their Photophysics and Cytotoxicity." Maisuls, I. et al., Inorganic Chemistry 2022, 61, 9195.
23. "Targeted siRNA nanocarrier: a platform technology for cancer treatment." Bäumer, N. et al., Oncogene 2022, 41, 2210
22. "Mapping the regioisomeric space and visible color range of purely organic dual emitters with ultralong phosphorescence components: From violet to red towards pure white-light." Roy, B. et al., Angewandte Chemie International Edition 2022, 61, e202111805
21. "AIE-Active Difluoroboron Complexes with N,O-Bidentate Ligands: Rapid Construction by Copper-Catalyzed." C−H Activation. Tan, G. et al., Advanced Science 2021, 8, 2101814
20. "Conjugated Pt(II) Complexes as Luminescence-Switch-On Reporters Addressing the Microenvironment of Bacterial Biofilms." Maisuls, I. et al., Inorganic Chemistry 2021, 60, 11058
19. "Intermolecular Interactions and Self-Assembly in Pt(II) Complex−Nanoclay Hybrids as Luminescent Reporters for Spectrally Resolved PLIM." Gangadharappa, S. C. et al., Journal of Physical Chemistry C 2021, 125, 5739
18. "Ligand-controlled and nanoconfinement-boosted luminescence employing Pt(II) and Pd(II) complexes: from color-tunable aggregation-enhanced dual emitters towards self-referenced oxygen reporters." Maisuls, I. et al., Chemical Science 2021, 12, 3270
17. "Compensation of Hybridization Defects in Phosphorescent Complexes with Pnictogen-Based Ligands - A Structural, Photophysical, and Theoretical Case-Study with Predictive Character." Gangadharappa, S. C. et al., Journal of the American Chemical Society 2020, 142, 21353
16. "Synthesis of Small-Molecule Fluorescent Probes for the In Vitro Imaging of Calcium-Activated Potassium Channel KCa3.1." Brömmel, K. et al., Angewandte Chemie International Edition 2020, 59, 8277
15. "A platinum-doped dendrimer as a phosphorescent label for bacteria in two-photon excitation microscopy." Molina Cabeza, N. et al., E. ACS Omega 2019, 4, 13027
14. "On-Surface Reactive Planarization of Pt(II) Complexes." Ren, J. et al., Angewandte Chemie International Edition 2019, 58, 15396
13. "Phosphorescent Pt(II) complexes spatially arrayed in micellar polymeric nanoparticles providing dual readout for multimodal imaging." Proetto, M. T. et al., Chemical Communications 2019, 55, 501
12. "Towards Optimized Naphthalocyanines as Sonochromes for Photoacoustic Imaging in vivo." Duffy, M. J. et al., Photoacoustics 2018, 9, 49
11. "Modification of the Potential Landscape of Molecular Rotors on Au(111) by the Presence of an STM Tip." Lu, H. et al., Nano Letters 2018, 18, 4704
10. "Oxygen-Insensitive Aggregates of Pt(II) Complexes as Phosphorescent Labels of Proteins with Luminescence Lifetime-Based Readouts." Delcanale, P. et al., ACS Applied Materials & Interfaces 2018, 10, 24361
9. "Quantitative assessment of intermolecular interactions by atomic force microscopy imaging using copper oxide tips." Mönig, H. et al., Nature Nanotechnology 2018, 13, 371
8. "Toward Tunable Electroluminescent Devices by Correlating Function and Submolecular Structure in 3D Crystals, 2D-Confined Monolayers, and Dimers." Wilde, S. et al., ACS Applied Materials & Interfaces 2018, 10, 22460
7. "Labeling and Selective Inactivation of Gram-Positive Bacteria Employing Bimodal Photoprobes with Dual Readouts." Galstyan, A. et al., Chemistry a European Journal 2016, 22, 5243
6. "Silicon(IV) Phthalocyanine-Decorated Cyclodextrin Vesicles as a Self-Assembled Phototherapeutic Agent against MRSA." Galstyan, A. et al., ACS Applied Materials & Interfaces 2016, 8, 12631
5. "Spatiotemporally Resolved Tracking of Bacterial Responses to ROS-Mediated Damage at the Single-Cell Level with Quantitative Functional Microscopy." Barroso Peña, A. et al., ACS Applied Materials & Interfaces 2016, 8, 15046
4. "Selective inactivation of resistant Gram-positive pathogens with a light-driven hybrid nanomaterial." Grüner, M. C.; et al., ACS Applied Materials & Interfaces 2015, 7, 20965
3. "Photofunctional surfaces for quantitative fluorescence microscopy: Monitoring the effects of photogenerated ROS at single cell level with spatiotemporal resolution." Stegemann, L. et al., ACS Applied Materials & Interfaces 2015, 7, 5944
2. "Scanning-Tunneling-Spectroscopy-Directed Design of Tailored Deep-Blue Emitters." Sanning, J. et al., Angewandte Chemie International Edition 2015, 54, 786
1. "Unraveling orbital hybridization of triplet emitters at the metal-organic interface." Ewen, P. et al., Physical Review Letters 2013, 111, 267401
